
Feb. 8, 2019
Research Highlight
Controlling the Length of Supramolecular Polymers
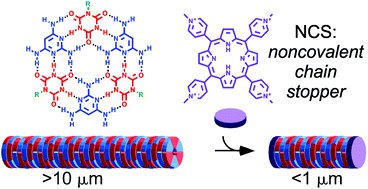
Supramolecular polymers are a kind of molecule constructed from smaller parts (monomers) that are not directly bonded to each other. Researchers found a way to control the length of these polymers in aqueous solution.Image credit: Karunakaran et al. 2018.
A new study has identified small molecules that can control the length of supramolecular polymers in aqueous solution. Supramolecular polymers are a kind of molecule constructed from smaller parts (monomers) that are not directly bonded to each other. Instead, the monomers of these large molecules are bound together through non-covalent bonds, which are reversible. This gives supramolecular polymers a number of unique traits, such as the ability to ‘self-heal.’ These properties make supramolecular polymers useful in a number of applications, including biomedical science.
A team of researchers has discovered a way to control the length of supramolecular polymers by using small molecules that act as noncovalent chain stoppers (NCSs). The supramolecular polymer used in the study (2,4,6-triaminopyrimidine) assembles in a way similar to DNA, and can form extremely long chains of monomers. The polymers form a hydrogel, and researchers were able to ‘tune’ the properties of this gel by adding NCSs to the system.
The study, “Exquisite regulation of supramolecular equilibrium polymers in water: chain stoppers control length, polydispersity and viscoelasticity,” was published in the journal Polymer Chemistry. The work was supported by NASA Astrobiology through the Exobiology Program and the NSF/NASA Center for Chemical Evolution (CCE) at the Georgia Institute of Technology in Atlanta, Georgia. The CCE is a collaborative program supported by the National Science Foundation (NSF) and the NASA Astrobiology Program.