
Nov. 9, 2018
Research Highlight
Naturally Occurring 'Batteries' Fueled Organic Carbon Synthesis on Mars
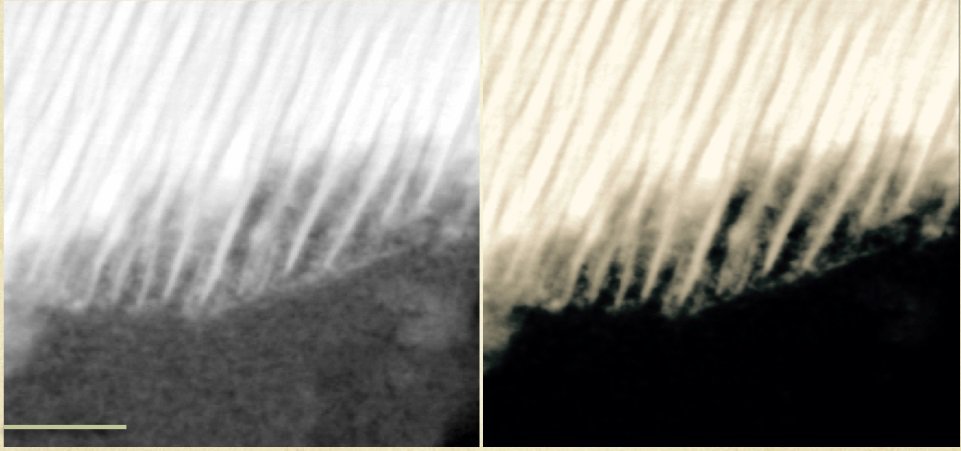
A high-resolution Transmission Electron Micrograph (scale 50nm) of a grain from a martian meteorite. Reminiscent of a long dinner fork, the organic carbon layers are found between the intact "tines." This texture is created when the volcanic minerals of the martian rock interact with a salty brine and become the anode and cathode of a naturally occurring battery in a corrosion reaction. This reaction would then have enough energy—under certain conditions—to synthesize organic carbon. Image courtesy of Andrew Steele.Image credit: Steele et al. (2018).
A new study in Science Advances describes how the interaction of martian rock and salty brine could lead to naturally occurring ‘batteries’ that would be able to sythesize organic carbon under certain conditions.
From the Carnegie Institution for Science:
Mars’ organic carbon may have originated from a series of electrochemical reactions between briny liquids and volcanic minerals, according to new analyses of three Martian meteorites from a team led by Carnegie’s Andrew Steele published in Science Advances.The group’s analysis of a trio of Martian meteorites that fell to Earth—Tissint, Nakhla, and NWA 1950—showed that they contain an inventory of organic carbon that is remarkably consistent with the organic carbon compounds detected by the Mars Science Laboratory’s rover missions.
In 2012, Steele led a team that determined the organic carbon found in 10 Martian meteorites did indeed come from the Red Planet and was not due to contamination from Earth, but also that the organic carbon did not have a biological origin. This new work takes his research to the next step—trying to understand how Mars’ organic carbon was synthesized, if not by biology.
Organic molecules contain carbon and hydrogen, and sometimes include oxygen, nitrogen, sulfur, and other elements. Organic compounds are commonly associated with life, although they can be created by non-biological processes as well, which are referred to as abiotic organic chemistry.
“Revealing the processes by which organic carbon compounds form on Mars has been a matter of tremendous interest for understanding its potential for habitability,” Steele said.
He and his co-authors took a deep dive into the minerology of these three Martian meteorites. Using advanced microscopy and spectroscopy, they were able to determine that the meteorites’ organic compounds were likely created by electrochemical corrosion of minerals in Martian rocks by a surrounding salty liquid brine.
“The discovery that natural systems can essentially form a small corrosion-powered battery that drives electrochemical reactions between minerals and surrounding liquid has major implications for the astrobiology field,” Steele explained.
Read the full article from the Carnegie Institution for Science.
The research was supported in part by the Astrobiology Science and Technology for Exploring Planets (ASTEP) element of the NASA Astrobiology Program. ASTEP was an active program element from 2001 to 2014 and supported investigations focused on exploring Earth’s extreme environments to learn how best to search for life on other planets. The types of projects that were funded by ASTEP are now competed under Planetary Science and Technology from Analog Research (PSTAR).
Additional support came from the Swedish National Space Board, the Swedish Research Council, the Helmholtz Recruiting Initiative, and the Department of Energy’s Office of Basic Energy Sciences.