2014 Annual Science Report
 NASA Ames Research Center
Reporting | SEP 2013 – DEC 2014
NASA Ames Research Center
Reporting | SEP 2013 – DEC 2014
Executive Summary
The Ames Team of the NAI investigates the physical, chemical and biological processes that combined to create early habitable environments. We trace the cosmic evolution of organic molecules from the interstellar medium, through protoplanetary disks and planetesimals, and ultimately to potentially habitable planets. We characterize the diversity of planetary systems that might emerge from protoplanetary disks. We identify diverse scenarios for the origins and early evolution of catalytic functionality and metabolic reaction networks. We develop and test a methodology for assessing quantitatively the habitability of early planetary environments – particularly Mars – via capabilities that could be deployed in situ. Our ongoing active involvement in multiple NASA missions provides context, incentives and collaborative opportunities for our research and education and public outreach programs. See www.amesteam.arc.nasa.gov.
Cosmic distribution of chemical complexity. We explore the connections between chemistry in space and the origins of life. We track the formation and evolution of chemical complexity in space, from simple carbon-rich molecules such as formaldehyde and acetylene to complex species including amino acids, nucleic acids and polycyclic aromatic hydrocarbons. We characterize carbon-rich species that are interesting biologically and we explore their possible roles in the origins of life on habitable worlds. We do this by measuring the spectra and chemistry of analog materials in the laboratory, by remote sensing with small spacecraft, and by analyses of extraterrestrial samples returned by spacecraft or that fall to Earth as meteorites. We use these results to interpret astronomical observations made with ground-based and orbiting telescopes.
We continued to study the chemistry that happens in space and the possibility that the organic products of this chemistry may be incorporated into forming planets and play a role in any subsequent origin of life. This work has focused on (i) radiation-induced chemistry that happens in astrophysical mixed-molecular ices of the types found in interstellar clouds and protostellar disks, and (ii) polycyclic aromatic hydrocarbons (PAHs), which are one of the main forms of molecular carbon in the universe.
The study of ice irradiation chemistry has focused on two, largely unrelated, issues. First, we conducted numerous simulations to study the radiation-induced chemistry that can occur in Pluto ice analogs when they are dosed with UV photons or electrons. Ices on Pluto are dominated by N2 but also contain small amounts of CO, CO2, and CH4. We demonstrated that irradiation of these ices leads to the formation of more complex organics that contain carboxylic acids, nitriles, and urea. These results are of interest in their own right but also because infrared spectra of these samples will help to interpret data returned from Pluto by the New Horizons spacecraft when it flies by Pluto later this year.
The other main task has been the completion of our work on studying the formation in ices of the pyrimidine-based nucleobases and other heterocyclic molecules. We demonstrated that all three of the pyrimidine-based nucleobases – uracil, cytosine, and thymine – are made when pyrimidine is irradiated in mixed-molecular ices.
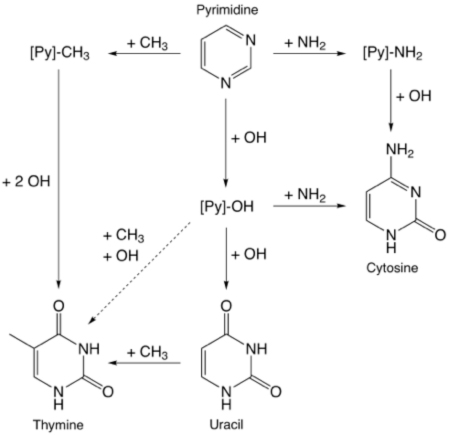
Interestingly uracil and cytosine are fairly easy to make, while the efficiency of production of thymine is much lower. It has been posited that the origin and evolution of life passed through an RNA World stage that preceded the use of DNA. Our results may provide an explanation of why this might have occurred – uracil may have been much more abundantly available than thymine in Earth’s early history. In related work, we showed that ice photochemistry can substitute O or N for skeletal C atoms in PAHs, i.e., PAHs irradiated in ices can be converted into O- and N-heterocycles. This process may form molecules like pyrimidine from which the nucleobases are formed.
This past year’s work on PAHs has focused on (i) interactions of PAHs with water ice as well as their photo-induced chemical reactions, and (ii) interpreting Spitzer observations using the PAH database. We recently published papers describing the impact PAH:water ice concentrations have on the resulting photo-chemistry. We have also explored the interactions between aromatic molecules, such as PAHs, and mineral substrates as well as any UV induced catalytic effects. We published the initial portion of this work in Langmuir.

This article was awarded the cover of this prestigious journal and was selected as the American Chemical Society Editor’s Choice article for the month of its publication. Also, we released a significantly updated version of the NASA Ames PAH IR Spectroscopic Database that i) includes very large PAHs, ii) allows users to import their astronomical spectra and perform least squares fitting analyses, and iii) quantifies the traditional qualitative proxy used to evaluate PAH charge distribution in diverse astronomical regions.
Evolution of protoplanetary disks and early planetary environments. We explore evolving protoplanetary disks to understand the formation of primitive planetary bodies that ultimately could host habitable environments. The disk is in many ways an astrochemical “primeval soup” in which abundant elements are assembled into increasingly complex organic compounds and mixed in the disk’s dust and gas envelope. We also investigate planetary processes that can influence early habitable environments.
Models of planet formation have shown that giant planets have a large impact on the number, masses, and orbits of terrestrial planets that form. They also play an important role in delivering volatiles from material that formed exterior to the snow line (the region in the disk beyond which water ice can condense) to the inner region of the disk where terrestrial planets can maintain liquid water on their surfaces. We presented simulations of the late stages of terrestrial planet formation from a disk of protoplanets around a solar-type star and we included a massive planet (from 1 M⊕ to 1 MJ ) in Jupiter’s orbit at ∼ 5.2 AU in all but one set of simulations. We examined two initial disk models with the same mass distribution and total initial water content, but with different distributions of water content. We compared the accretion rates and final water mass fraction of the planets that form. Remarkably, all of the planets that formed in our simulations without giant planets were water-rich, showing that giant planet companions are not required to deliver volatiles to terrestrial planets in the habitable zone. In contrast, an outer planet at least several times the mass of Earth may be needed to clear distant regions of debris truncating the epoch of frequent large impacts. Observations of exoplanets from radial velocity surveys suggest that outer Jupiter-like planets may be scarce therefore our findings indicate that there might be more habitable planets residing in our galaxy than previously thought.
Mineralogical Traces of Early Habitable Environments. We seek to understand how habitability (potential to support life) varies across a range of physical and chemical parameters, in order to support a long-term goal of characterizing habitability of environments on Mars. We are examining the interplay between physicochemical environment and associated microbial communities in a subsurface environment dominated by serpentinization (a reaction involving water and crustal rocks, which indicated by surface mineralogy to have occurred on ancient Mars). We are determining how mineral assemblages become a lasting record of prior environmental conditions, and therefore indicate prior habitability. This supports interpretations of mineralogy data obtained by the CheMin instrument on the Mars Science Laboratory.
We focused on the serpentinite borehole observatories at McLaughlin Natural Reserve, established in 2011. Samples for aqueous geochemistry and molecular biology were collected and analyzed quarterly from the boreholes as part of a seasonal monitoring campaign. We conducted an extensive metagenomic and pyrotag sequencing analysis of fluids and solid material from the new wells and compared these against in situ metabolic activity assays. We completed sequencing and analysis of metagenomes from cored material and recovered well fluids, X-ray diffraction analyses of cored materials, and 18 months of seasonal monitoring. We installed and recovered a first round of down-hole microbial colonization experiments and conducted an initial suite of enrichment culturing experiments. We published in Frontiers in Microbiology and Scientific Drilling, and presented at the Tenth International Symposium on Subsurface Microbiology and the AGU Fall Meeting.
Origins of functional proteins and the early evolution of metabolism. We seek to identify critical requirements for the emergence of biological complexity in early habitable environments by examining key steps in the origins and early evolution of functional proteins and metabolic reaction networks. We investigate whether protein functionality can arise from an inventory of polypeptides that might have naturally existed in habitable environments, and we investigate how primordial proteins could evolve through the diversification of their structure and function and determine how simple proteins could carry out seemingly complex functions that are essential to life. This work offers unique information about the earliest evolution of cellular systems that has not been available from other studies.
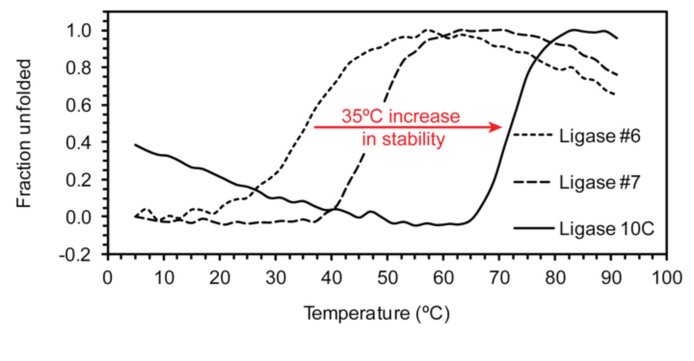
We continued investigations of model primordial enzymes. One approach to creating such enzymes is to introduce mutations into an existing protein to enable a new catalytic activity. Ideally, enzymatic activity and protein stability are engineered simultaneously to ensure that isolated enzymes are both stable and have the desired catalytic properties. We use the in vitro selection technique mRNA display to achieve both in a single step. Starting with a library of artificial RNA ligase enzymes that were mostly mesophilic, we selected for thermostable active enzyme variants by performing the selection step at 65 °C. The most efficient enzyme, ligase 10C, was not only active at 65 °C, but was also an order of magnitude more active at room temperature compared to related enzymes previously isolated at ambient temperature.
Concurrently, the melting temperature of ligase 10C increased by 35 degrees compared to these related enzymes. These results highlight the versatility of the in vitro selection technique mRNA display as a powerful method for the isolation of thermostable models of primordial enzymes.
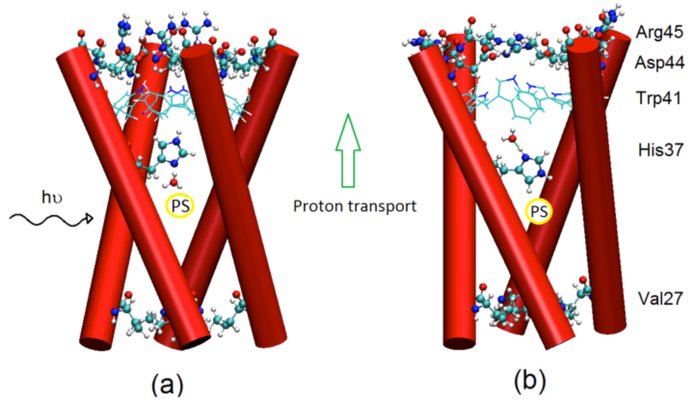
Another essential function of primordial proteins was transmembrane proton transfer that facilitated transduction of energy for metabolic functions. The reliable, efficient and controlled generation of proton gradients became possible only with the emergence of active proton pumps. We studied a small, hexameric, transmembrane M2 proton channel from influenza A virus as a model for proton transport. Remarkably, the M2 channel and its truncated version built of only 24 residues per monomer contain almost all mechanistic features of markedly larger, modern proton pumps. Nδ in the imidazole rings of the His37 sensor can be considered as the primary acceptor that receives a proton from the aqueous cavity near Gly34. In response to protonation, histidine undergoes conformational change shuttling the proton to the opposite side of the sensor. Deprotonation on the outgoing side is followed by the return of histidine to its initial conformation preventing back transfer of the proton to the cavity. Trp41 and Asp44 also ensure directional proton transfer to the aqueous solution on the outgoing side. The Val27 gate protects protons that enter the cavity from returning to the incoming side. It appears that the only missing element that would turn M2 to a proton pump is a proton source that would be located near the aqueous cavity.
PAH molecules that are sensitive to ultraviolet light might serve as sources. PAHs are believed to be abundant on the early earth and have been proposed to provide protons for protocellular functions. The smallest PAH, naphthalene, is comparable in size to amantadine, a drug known to reside in the Gly34 cavity. M2 is not an evolutionarily early protein but its structure and mechanism of action indicates that simple proteins that conceivably existed early in the protobiological era might have captured the main features of proton pumps. Such proteins might have been at the roots of active proton transport.
Education and Public Outreach. Ames Team members pursued activities in education and public outreach (EPO) that included efforts in high-impact public venues. Team research efforts have contributed source material for the team’s EPO program. We administered a graduate-level astrobiology course at the University of California at Santa Cruz. We conducted lab tours and demonstrations for students and educators. We lectured to K-12 classrooms, universities, museums, planetariums, national park visitor centers, scientific conferences and the public media. Team members worked with staff at Red Bluff High School and Lassen Volcanic National Park to offer the Lassen Astrobiology Student Intern Program to high school juniors and seniors. Each intern examined hydrothermal features, collected field data and wrote a report that interpreted hydrothermal field sites and geochemical and biological analyses. Ames team members trained Lassen Park interpretive staff and worked with them to develop trailside signs located at park sites that illustrate astrobiology research. The team contributed substantially to the three-day 2014 Lassen Dark Sky Festival by delivering six lectures in the visitor center and staffing a NASA booth.
Mission involvement. The team maintained its substantial presence in NASA missions. The P.I. is a member of the Mars Exploration Rover (MER) mission, the Mars Reconnaissance Orbiter (MRO) CRISM team and the Mars Science Laboratory (MSL) mission CheMin team. The P.I. served as Chair of MEPAG (NASA’s Mars Exploration Program Analysis Group) for a third year. A Co-I is P.I. of the MSL Curiosity CheMin team. Another Co-I is the PI on a Comet Surface Sample Return mission concept for the next New Frontiers AO. Other team members participate in the following: Kepler, Stardust, Stratospheric Observer for Infrared Astronomy, Mars Reconnaissance Orbiter, MSL, James Webb Space Telescope, Spitzer Space Telescope, Herschel, O/OREOS, ABE/ASPIRE, LCROSS, LADEE, and the Comet Coma Rendezvous Sample Return mission.
Publications
-
A. Wilson, M., Wei, C., & Pohorille, A. (2014). Towards Co-Evolution of Membrane Proteins and Metabolism. Orig Life Evol Biosph, 44(4), 357–361. doi:10.1007/s11084-014-9393-2
-
Cook, A. M., Mattioda, A. L., Quinn, R. C., Ricco, A. J., Ehrenfreund, P., Bramall, N. E., … Walker, R. (2014). SEVO ON THE GROUND: DESIGN OF A LABORATORY SOLAR SIMULATION IN SUPPORT OF THE O/OREOS MISSION. The Astrophysical Journal Supplement Series, 210(2), 15. doi:10.1088/0067-0049/210/2/15
-
Cook, A. M., Mattioda, A. L., Ricco, A. J., Quinn, R. C., Elsaesser, A., Ehrenfreund, P., … Hoffmann, S. V. (2014). The Organism/Organic Exposure to Orbital Stresses (O/OREOS) Satellite: Radiation Exposure in Low-Earth Orbit and Supporting Laboratory Studies of Iron Tetraphenylporphyrin Chloride. Astrobiology, 14(2), 87–101. doi:10.1089/ast.2013.0998
-
Cook, A. M., Ricca, A., Mattioda, A. L., Bouwman, J., Roser, J., Linnartz, H., … Allamandola, L. J. (2015). PHOTOCHEMISTRY OF POLYCYCLIC AROMATIC HYDROCARBONS IN COSMIC WATER ICE: THE ROLE OF PAH IONIZATION AND CONCENTRATION. The Astrophysical Journal, 799(1), 14. doi:10.1088/0004-637x/799/1/14
-
Crespo-Medina, M., Twing, K. I., Kubo, M. D. Y., Hoehler, T. M., Cardace, D., McCollom, T., & Schrenk, M. O. (2014). Insights into environmental controls on microbial communities in a continental serpentinite aquifer using a microcosm-based approach. Frontiers in Microbiology, 5. doi:10.3389/fmicb.2014.00604
-
Elsaesser, A., Quinn, R. C., Ehrenfreund, P., Mattioda, A. L., Ricco, A. J., Alonzo, J., … Santos, O. (2014). Organics Exposure in Orbit (OREOcube): A Next-Generation Space Exposure Platform. Langmuir, 30(44), 13217–13227. doi:10.1021/la501203g
-
Hoehler, T. M., & Alperin, M. J. (2014). Biogeochemistry: Methane minimalism. Nature, 507(7493), 436–437. doi:10.1038/nature13215
-
Houghton, J., Fike, D., Druschel, G., Orphan, V., Hoehler, T. M., & Des Marais, D. J. (2014). Spatial variability in photosynthetic and heterotrophic activity drives localized δ 13 C org fluctuations and carbonate precipitation in hypersaline microbial mats. Geobiology, 12(6), 557–574. doi:10.1111/gbi.12113
-
Lee, J. Z., Burow, L. C., Woebken, D., Everroad, R. C., Kubo, M. D., Spormann, A. M., … Hoehler, T. M. (2014). Fermentation couples Chloroflexi and sulfate-reducing bacteria to Cyanobacteria in hypersaline microbial mats. Frontiers in Microbiology, 5. doi:10.3389/fmicb.2014.00061
-
Materese, C. K., Cruikshank, D. P., Sandford, S. A., Imanaka, H., Nuevo, M., & White, D. W. (2014). ICE CHEMISTRY ON OUTER SOLAR SYSTEM BODIES: CARBOXYLIC ACIDS, NITRILES, AND UREA DETECTED IN REFRACTORY RESIDUES PRODUCED FROM THE UV PHOTOLYSIS OF N 2 :CH 4 :CO-CONTAINING ICES. The Astrophysical Journal, 788(2), 111. doi:10.1088/0004-637x/788/2/111
-
Mattioda, A. L., Bauschlicher, C. W., Bregman, J. D., Hudgins, D. M., Allamandola, L. J., & Ricca, A. (2014). Infrared vibrational and electronic transitions in the dibenzopolyacene family. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 130, 639–652. doi:10.1016/j.saa.2014.04.017
-
Morelli, A., Haugner, J., & Seelig, B. (2014). Thermostable Artificial Enzyme Isolated by In Vitro Selection. PLoS ONE, 9(11), e112028. doi:10.1371/journal.pone.0112028
-
Quintana, E. V., & Lissauer, J. J. (2014). THE EFFECT OF PLANETS BEYOND THE ICE LINE ON THE ACCRETION OF VOLATILES BY HABITABLE-ZONE ROCKY PLANETS. The Astrophysical Journal, 786(1), 33. doi:10.1088/0004-637x/786/1/33
-
Rosenberg, M. J. F., Berné, O., & Boersma, C. (2014). Random mixtures of polycyclic aromatic hydrocarbon spectra match interstellar infrared emission. A&A, 566, L4. doi:10.1051/0004-6361/201423953
-
Saha, R., Pohorille, A., & Chen, I. A. (2014). Molecular Crowding and Early Evolution. Orig Life Evol Biosph, 44(4), 319–324. doi:10.1007/s11084-014-9392-3
-
Sandford, S. A., Bera, P. P., Lee, T. J., Materese, C. K., & Nuevo, M. (2014). Photosynthesis and Photo-Stability of Nucleic Acids in Prebiotic Extraterrestrial Environments. Topics in Current Chemistry, None, 123–164. doi:10.1007/128_2013_499
-
Wei, C., & Pohorille, A. (2015). M2 Proton Channel: Toward a Model of a Primitive Proton Pump. Orig Life Evol Biosph, 45(1-2), 241–248. doi:10.1007/s11084-015-9421-x
-
Wilson, M. A., Nguyen, T. H., & Pohorille, A. (2014). Combining molecular dynamics and an electrodiffusion model to calculate ion channel conductance. J. Chem. Phys., 141(22), 22D519. doi:10.1063/1.4900879
-
Yabuta, H., Uesugi, M., Naraoka, H., Ito, M., Kilcoyne, A. L. D., Sandford, S. A., … Abe, M. (2014). X-ray absorption near edge structure spectroscopic study of Hayabusa category 3 carbonaceous particles. Earth Planet Sp, 66(1), None. doi:10.1186/s40623-014-0156-0
-
Yada, T., Fujimura, A., Abe, M., Nakamura, T., Noguchi, T., Okazaki, R., … Kawaguchi, J. (2013). Hayabusa-returned sample curation in the Planetary Material Sample Curation Facility of JAXA. Meteoritics & Planetary Science, 49(2), 135–153. doi:10.1111/maps.12027