2013 Annual Science Report
 NASA Goddard Space Flight Center
Reporting | SEP 2012 – AUG 2013
NASA Goddard Space Flight Center
Reporting | SEP 2012 – AUG 2013
Analysis of Prebiotic Organic Compounds in Astrobiologically Relevant Samples
Project Summary
The Astrobiology Analytical Laboratory (AAL) of the GCA is dedicated to the study of organic compounds derived from past and future sample return missions, meteorites, lab simulations of Mars, interstellar, proto-planetary, and cometary ices and grains, and instrument development. This year, we analyzed the amino acid and nucleobase content of a martian meteorite; our findings suggested the presence of extraterrestrial amino acids in that meteorite. We studied irradiated benzene ices to determine that this type of radiation chemistry may have produced some of the complex aromatics found in meteorites. We identified amino acids for the first time in high-metal carbonaceous chondrite classes, supporting the idea of multiple formation mechanisms for these astrobiologically relevant compounds. We supported development of a liquid chromato-graphmass spectrometer aimed at in situ analyses of amino acids and chirality on airless bodies including asteroids and the outer planet’s icy moons Enceladus and Europa. We hosted a graduate student, an undergraduate, and a high-school intern, and participated in numerous public outreach and education events. We continued our participation in the OSIRIS-REx asteroid sample return mission and provided support for the Sample Analysis at Mars instrument of NASA’s Mars rover Curiosity.
Project Progress
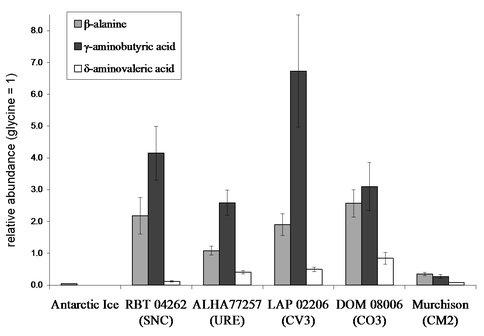
We analyzed the amino acid and nucleobase content of the shergottite Roberts Massif (RBT) 04262 using liquid chromatography-mass spectrometry (LC-MS). We did not detect any nucleobases above our detection limit in formic acid extracts; however, we did measure a suite of protein and non-protein amino acids in hot-water extracts. The distribution of these amino acids matched those we previously measured from thermally altered carbonaceous chondrites (Figure 1). While previous results strongly indicated that most if not all of the amino acids in Martian meteorites were the result of terrestrial contamination, RBT 04642 provided the first reasonable indication of extraterrestrial amino acids in a Martian meteorite.
We reported new studies of the organic residue made from benzene irradiated at 19 K by 0.8 MeV protons. Atmospheric pressure photoionization coupled to high resolution Orbitrap mass spectrometry was used to determine molecular composition, and accurate mass measurements suggested the presence of complex aromatic compounds. Our results are consistent with the possibility that solid phase radiation chemistry of benzene produced some of the complex aromatics found in meteorites. Furthermore, radiation-induced reactions provide a facile mechanism for the extensive hydrogenation (likely) observed in meteoritic organics.
We published our analysis of six carbonaceous chondrites from the metal-rich CH and CB classes that had not previously been investigated for amino acids. We made the first reported discovery of amino acids in these meteorites, using enantiomeric and carbon isotopic measurements to support their extraterrestrial origin. We observed distinct differences in their amino acid distributions, providing evidence that multiple mechanisms were active in forming amino acids in CH and CB chondrites.
As part of a large meteorite research consortium, we published the results of our analysis of three fragments of the Sutter’s Mill meteorite, a CM carbonaceous chondrite that fell in California in April 2012. We used our optimized analytical techniques to examine the amino acid content and bulk carbon and nitrogen isotopic ratios for these samples. The low abundances of amino acids found in the Sutter’s Mill samples compared to other CM2 meteorites was consistent with other measurements of Sutter’s Mill indicating that the meteorite’s parent body had experienced significant heating, up to 300°C prior to entry.
We continued to explore crystallization structures of amino acids and the possible role of these structures in amplifying enantiomeric excesses of meteoritic amino acids. We published the crystal structures of two such compounds.
Our NAI NPP postdoctoral fellow (José Aponte) developed new methods for the detection and isotopic analysis of meteoritic amines for comparison with meteoritic amino acids. We expect to submit our analyses of the Murchison meteorite for publication shortly.
We supported the ASTID-funded development of a miniaturized LC-MS for in-situ analyses of amino acids and other soluble organics on icy planetary surfaces (ASTID PI Stephanie Getty). Our state-of-the-art laboratory equipment was used for component testing and method development.
We hosted one graduate student, one undergraduate, and one high-school intern. Graduate student Karen Smith, from the Penn State NAI team, completed her work in our lab examining nitrogen heterocyclic molecules in spark-discharge experiments and in meteorites. Undergraduate Jolie Carlisle (University of California at Berkeley) assisted with the meteoritic amine method development during her appointment under the GCA’s URAA summer program, while high-school student Sarah Grunsfeld (Clarksville, MD) worked on the analysis of meteoritic amino acids and amino acid crystallizations.
Dworkin continued to serve as Project Scientist for the OSIRIS-REx New Frontiers-3 asteroid sample return mission, which addresses the Astrobiology Roadmap goal of determining chemical precursors of life in the solar system. Glavin is a Co-I, while Elsila and Callahan are Collaborators. The Astrobiology Analytical Lab is heavily involved in this mission, both in the Contamination Control and Contamination Knowledge areas, radiation experiments to understand the effect of space weathering on amino acids and other organics in meteorites, and in the eventual analysis of returned asteroidal material. The infrastructure, in part provided by NAI funding, continues to be used in OSIRIS-REx analysis in support of the development of an innovative contamination control approach to preserving chemistry important to astrobiology under a limited budget. This approach has been praised by independent science and engineering reviewers and is likely to be the template for contamina-tion control for Mars sample return.
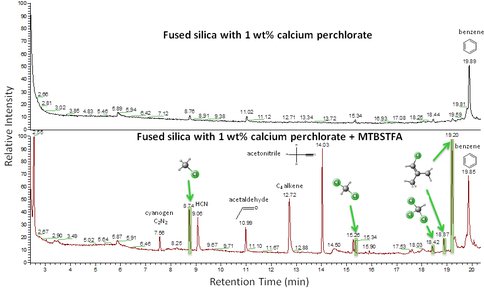
As part of Glavin’s Mars Science Laboratory Participating Scientist Program study, we sup-ported the Sample Analysis at Mars (SAM) instrument data analysis effort. We performed laboratory pyrolysis gas chromatography mass spectrometry experiments on a variety of terrestrial Mars analogs and synthetic standards to better understand the origin of the chlo-rinated hydrocarbons detected by the SAM instrument at the Rocknest aeolian drift in Gale Crater. Experiments using AAL instrumentation performed by MSL PSP Project Collabora-tor’s Millie Martin and Caroline Freissinet provided a critical set of data that demonstrated that the carbon in the chlorinated hydrocarbons detected at Rocknest most likely originated from MTBSTFA, one of the wet chemistry reagents used in SAM (Fig. 2). However, the chlo-rine was derived from martian oxychlorine compounds, such as calcium perchlorate.
Publications
-
Brewer, G., Burton, A. S., Dworkin, J. P., & Butcher, R. J. (2013). 2-Methylaspartic acid monohydrate. Acta Cryst E Struct Rep Online, 69(12), o1856–o1857. doi:10.1107/s1600536813032170
-
Burton, A. S., Elsila, J. E., Hein, J. E., Glavin, D. P., & Dworkin, J. P. (2013). Extraterrestrial amino acids identified in metal-rich CH and CB carbonaceous chondrites from Antarctica. Meteoritics & Planetary Science, 48(3), 390–402. doi:10.1111/maps.12063
-
Butcher, R. J., Brewer, G., Burton, A. S., & Dworkin, J. P. (2013). Isovaline monohydrate. Acta Cryst E Struct Rep Online, 69(12), o1829–o1830. doi:10.1107/s1600536813031620
-
Callahan, M. P., Burton, A. S., Elsila, J. E., Baker, E. M., Smith, K. E., Glavin, D. P., & Dworkin, J. P. (2013). A search for amino acids and nucleobases in the Martian meteorite Roberts Massif 04262 using liquid chromatography-mass spectrometry. Meteoritics & Planetary Science, 48(5), 786–795. doi:10.1111/maps.12103
-
Callahan, M. P., Gerakines, P. A., Martin, M. G., Peeters, Z., & Hudson, R. L. (2013). Irradiated benzene ice provides clues to meteoritic organic chemistry. Icarus, 226(2), 1201–1209. doi:10.1016/j.icarus.2013.07.033
-
Glavin, D. P., Freissinet, C., Miller, K. E., Eigenbrode, J. L., Brunner, A. E., Buch, A., … Mahaffy, P. R. (2013). Evidence for perchlorates and the origin of chlorinated hydrocarbons detected by SAM at the Rocknest aeolian deposit in Gale Crater. Journal of Geophysical Research: Planets, 118(10), 1955–1973. doi:10.1002/jgre.20144
-
Jenniskens, P., Fries, M. D., Yin, Q-Z., Zolensky, M., Krot, A. N., Sandford, S. A., … Worden, S. P. (2012). Radar-Enabled Recovery of the Sutter’s Mill Meteorite, a Carbonaceous Chondrite Regolith Breccia. Science, 338(6114), 1583–1587. doi:10.1126/science.1227163
-
Leshin, L. A., Mahaffy, P. R., Webster, C. R., Cabane, M., Coll, P., Conrad, P. G., … Moores, J. E. (2013). Volatile, Isotope, and Organic Analysis of Martian Fines with the Mars Curiosity Rover. Science, 341(6153), 1238937–1238937. doi:10.1126/science.1238937
-
Parker, E. T., Cleaves, J. H., Burton, A. S., Glavin, D. P., Dworkin, J. P., Zhou, M., … Fernández, F. M. (2014). Conducting Miller-Urey Experiments. JoVE, None(83), None. doi:10.3791/51039
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Jason Dworkin
Project Investigator
Michael Callahan
Co-Investigator
Jamie Cook
Co-Investigator
Daniel Glavin
Co-Investigator
Jose Aponte
Collaborator
Aaron Burton
Collaborator
Caroline Freissinet
Collaborator
Mildred Martin
Collaborator
Jennifer Stern
Collaborator
-
RELATED OBJECTIVES:
Objective 2.1
Mars exploration.
Objective 3.1
Sources of prebiotic materials and catalysts