2009 Annual Science Report
 NASA Ames Research Center
Reporting | JUL 2008 – AUG 2009
NASA Ames Research Center
Reporting | JUL 2008 – AUG 2009
Cosmic Distribution of Chemical Complexity
Project Summary
This project seeks to improve our understanding of the connection between chemistry in space and the origin of life on Earth and possibly other worlds. Our approach is to trace the formation and development of chemical complexity in space, with particular emphasis on understanding the evolution from simple to complex species focusing on those that are interesting from a biogenic perspective and also understanding their possible roles in the origin of life on habitable worlds. We do this by first measuring the spectra and chemistry of materials under simulated space conditions in the laboratory. We then use these results to interpret astronomical observations made with ground-based and orbiting telescopes. We also carry out experiments on simulated extraterrestrial materials to analyze extraterrestrial samples returned by NASA missions or that fall to Earth in meteorites.
Project Progress
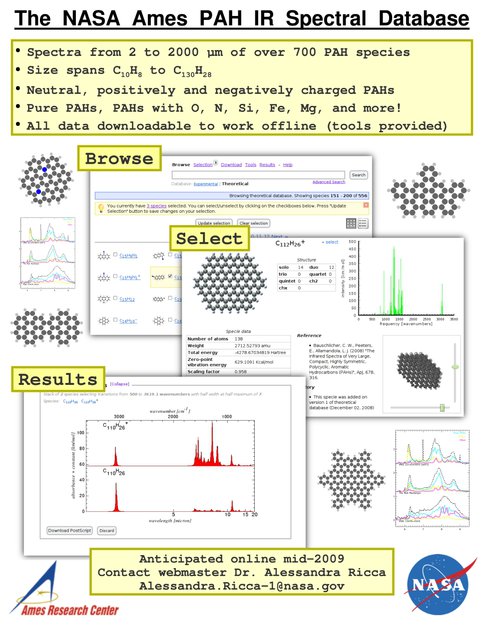
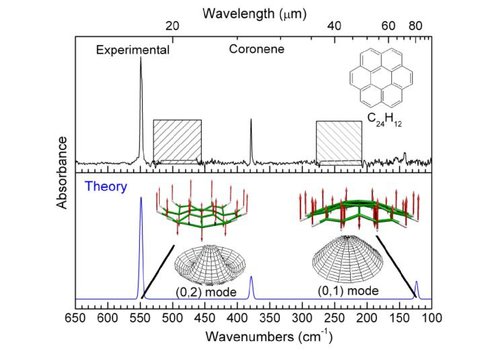
Our unique collection of polycyclic aromatic hydrocarbon (PAH) spectra is now in database form. Tools to query the data and analyze astronomical spectra are being finalized for web launch. (Figure 1) This will revolutionize how astronomers analyze PAH spectra and understand how carbonaceous species evolve across the universe. We published five PAH-related papers, as follows: 1- Far-IR (Figure 2) and 1- 15 to 20 µm spectra of PAHs in preparation for Herschel and SOFIA. 2- on mid-IR spectroscopy of large PAHs. 1- on photochemistry of the PAH pyrene in water analogs of interstellar and Solar System ices. This last paper opens a new field of research.
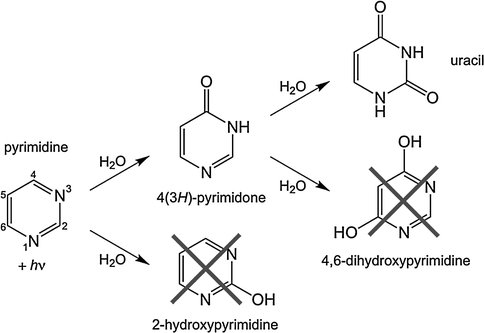
We have published one paper and are working on several others describing reactions that produce prebiotic compounds by UV irradiation of cosmic ices. The paper published in Astrobiology described experiments showing that the photolysis of pyrimidine in H2O ices produces a host of new compounds, one of which is the nucleobase uracil (Figure 3). A second paper is in preparation showing that the addition of ammonia to the ice results in the production of the nucleobase cytosine.
One of us (Sandford) continues to be intimately involved with extraction, distribution, and analysis of samples from Comet 81P/Wild 2 returned by the Stardust mission (Figure 4). He is also continues to work as a CoI on the Hayabusa asteroid sample return mission, which is due back to Earth in June 2010. Another team member (Mattioda) is on the Science Team for the O/OREOS (Organisms/ORganics Exposure to Orbital Stresses), NASA’s first Astrobiology Small Payloads mission (Figure 5). He and Bramall are working on the SEVO (Space Environment Viability of Organics) component for O/OREOS.
This year Pascale Ehrenfreund, Wisconsin team, in collaboration with Louis Allamandola and Andrew Mattioda was awarded an NAI DDF grant to investigate the modification of organic materials under interstellar conditions via UV-Visible spectroscopy, particularly polycyclic aromatic hydrocarbons (PAHs). This DDF employs a post-doc Kathryn Bryson. This past year Kathryn has setup the UV-Vis spectrometer system and begun the collection of spectra for thin films of astrobiologically-relevant organic molecules.
Figure 1. The NASA Ames PAH IR spectral database. The yellow highlighted box summarizes the spectroscopic range covered and the variety of species in the database. The lower web 'screen shots’ are examples of the tools and capabilities of our web site.
Figure 2. Experimental (top) and theoretical (bottom) FIR spectra of coronene
(C24H12). The cross hatched areas in the experimental spectrum correspond to regions that are not accessible with the 12.5 μm mylar beam splitter. The molecular structures and arrows show the vibrational modes that give rise to the bands. The actual atom displacements are exaggerated by about a factor of 10.
Figure 3. The UV photolysis of pyrimidine in H2O ices results in the production of the nucleobase uracil.
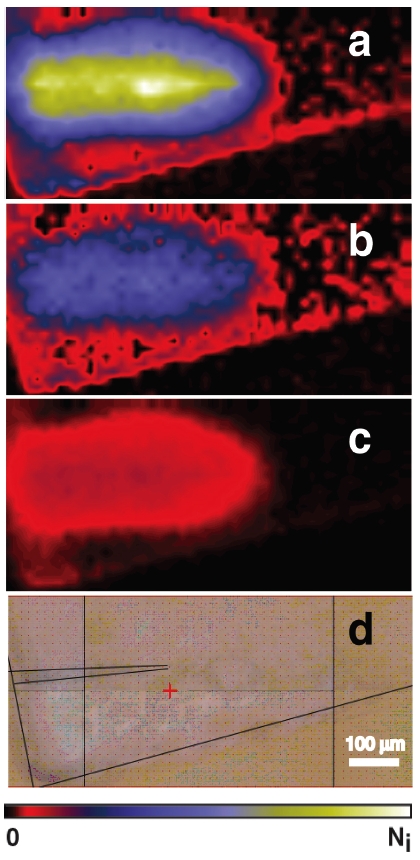
Figure 4. Infrared absorbance maps for (a) CH2, (b) -CH3, and© C=O compared to the optical image (d) of particle track C2009,4,59 made by impact of a sample from comet Wild 2 into Stardust aerogel. The same false color bar is used in all three absorbance maps. All four maps show identical area (same distance scale). The original aerogel surface exposed to the comet is on the left. These images show that the impacting comet particle contained aliphatic organic materials that were distributed into the aerogel surrounding the track during the impact event.
Figure 5. Computer-generated image of the complete O/OREOS nanosatellite. Right: photograph of the flight prototype. The wheel near the left end of the satellite is the SEVO sample carousel with 24 cells (that appear as round holes) for thin-film organic samples.
Publications
-
Bajt, S., Sandford, S. A., Flynn, G. J., Matrajt, G., Snead, C. J., Westphal, A. J., & Bradley, J. P. (2009). Infrared spectroscopy of Wild 2 particle hypervelocity tracks in Stardust aerogel: Evidence for the presence of volatile organics in cometary dust. Meteoritics & Planetary Science, 44(4), 471–484. doi:10.1111/j.1945-5100.2009.tb00745.x
-
Bauschlicher, C. W., Peeters, E., & Allamandola, L. J. (2009). THE INFRARED SPECTRA OF VERY LARGE IRREGULAR POLYCYCLIC AROMATIC HYDROCARBONS (PAHs): OBSERVATIONAL PROBES OF ASTRONOMICAL PAH GEOMETRY, SIZE, AND CHARGE. The Astrophysical Journal, 697(1), 311–327. doi:10.1088/0004-637x/697/1/311
-
Boersma, C., Bauschlicher, C. W., Allamandola, L. J., Ricca, A., Peeters, E., & Tielens, A. G. G. M. (2010). The 15–20 μ m PAH emission features: probes of individual PAHs?. A&A, 511, A32. doi:10.1051/0004-6361/200912714
-
Boersma, C., Mattioda, A. L., Bauschlicher, C. W., Peeters, E., Tielens, A. G. G. M., & Allamandola, L. J. (2008). THE 5.25 AND 5.7 μm ASTRONOMICAL POLYCYCLIC AROMATIC HYDROCARBON EMISSION FEATURES. The Astrophysical Journal, 690(2), 1208–1221. doi:10.1088/0004-637x/690/2/1208
-
Boersma, C., Mattioda, A. L., Bauschlicher, C. W., Peeters, E., Tielens, A. G. G. M., & Allamandola, L. J. (2009). ERRATUM: “THE 5.25 AND 5.7 μm ASTRONOMICAL POLYCYCLIC AROMATIC HYDROCARBON EMISSION FEATURES” (2009, ApJ, 690, 1208). The Astrophysical Journal, 694(1), 704–705. doi:10.1088/0004-637x/694/1/704
-
Bouwman, J., Paardekooper, D. M., Cuppen, H. M., Linnartz, H., & Allamandola, L. J. (2009). REAL-TIME OPTICAL SPECTROSCOPY OF VACUUM ULTRAVIOLET IRRADIATED PYRENE:H 2 O INTERSTELLAR ICE. The Astrophysical Journal, 700(1), 56–62. doi:10.1088/0004-637x/700/1/56
-
Glavin, D. P., Dworkin, J. P., & Sandford, S. A. (2008). Detection of cometary amines in samples returned by Stardust. Meteoritics & Planetary Science, 43(1-2), 399–413. doi:10.1111/j.1945-5100.2008.tb00629.x
-
Mattioda, A. L., Ricca, A., Tucker, J., Bauschlicher, C. W., & Allamandola, L. J. (2009). FAR-INFRARED SPECTROSCOPY OF NEUTRAL CORONENE, OVALENE, AND DICORONYLENE. The Astronomical Journal, 137(4), 4054–4060. doi:10.1088/0004-6256/137/4/4054
-
Nuevo, M., Milam, S. N., Sandford, S. A., Elsila, J. E., & Dworkin, J. P. (2009). Formation of Uracil from the Ultraviolet Photo-Irradiation of Pyrimidine in Pure H 2 O Ices. Astrobiology, 9(7), 683–695. doi:10.1089/ast.2008.0324
-
Sandford, S. A. (2008). Terrestrial Analysis of the Organic Component of Comet Dust*. Annual Review of Analytical Chemistry, 1(1), 549–578. doi:10.1146/annurev.anchem.1.031207.113108
- Cody, G.,.D., Ade, H., Alexander, C.M.O.D., Araki, T., Butterworth, A., Fleckenstein, H., Flynn, G., Gilles, M.K., Jacobsen, C., Kilcoyne, A.L.D., Messenger, K., Sandford, S.A., Tyliszczak, T., Westphal, A.J., Wirick, S. & Yabuta, H. (2008). Quantitative Organic and Light-Element Analysis of Comet 81P/Wild 2 Particles using C-, N-, and O- µ-XANES. Meteoritics and Planetary Science, 43: 353-365.
- Kwok, S. & Sandford, S. (2008). Organic Matter in Space. Proceedings of IAU. Vol. Proc. Of IAU Symposium 251. Cambridge University Press: Cambridge.
- Matrajt, G., Ito, M., Wirick, S., Messenger, S., Brownlee, D.E., Joswiak, D., Flynn, G., Sandford, S., Snead, C. & Westphal, A. (2008). Carbon Investigation of Two Stardust Particles: A TEM, NanoSIMS, and XANES Study. Meteoritics and Planetary Science, 43: 315-334.
- Rotundi, A., Baratta, G.A., Borg, J., Brucato, J.R., Busemann, H., Colangeli, L., D’HenDecourt, L., Djouadi, Z., Ferrini, G., Franchi, I.A., Fries, M., Grossemy, F., Keller, L.P., Mennella, V., Nakamura, K., Nittler, L.R., Palumbo, M.E., Sandford, S.A., Steele, A. & Wopenka, B. (2008). Combined Micro-Raman, Micro-Infrared, and Field Emission Scanning Electron Microscope Analyses of Comet 81P/Wild 2 Particles Collected by Stardust. Meteoritics and Planetary Science, 43: 367-397.
- Sandford, S.A. (2008). Organics in the Samples Returned from Comet 81P/Wild 2 by the Stardust Spacecraft. In: Kwok, S. & Sandford, S. (Eds.). Organic Matter in Space. Vol. Proc. Of IAU Symposium 251. Cambridge University Press: Cambridge.
- Yabuta, H., Cody, G.D., Kilcoyne, A.L.D., Araki, T., Alexander, C.M.O.D. & Sandford, S. (2009). X-ray absorption near edge spectroscopy analyses of organic matter in comet particles (in Japanese). Researches in Organic Geochemistry, 23: 147-157.
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Murthy Gudipati
Co-Investigator
Andrew Mattioda
Co-Investigator
Scott Sandford
Co-Investigator
Max Bernstein
Collaborator
Jan Cami
Collaborator
Jamie Cook
Collaborator
Jason Dworkin
Collaborator
Els Peeters
Collaborator
Nathan Bramall
Postdoc
Stefanie Milam
Postdoc
Michel Nuevo
Postdoc
Joseph Roser
Postdoc
-
RELATED OBJECTIVES:
Objective 1.1
Formation and evolution of habitable planets.
Objective 2.1
Mars exploration.
Objective 2.2
Outer Solar System exploration
Objective 3.1
Sources of prebiotic materials and catalysts
Objective 3.2
Origins and evolution of functional biomolecules
Objective 3.4
Origins of cellularity and protobiological systems
Objective 4.3
Effects of extraterrestrial events upon the biosphere
Objective 7.1
Biosignatures to be sought in Solar System materials
Objective 7.2
Biosignatures to be sought in nearby planetary systems