2008 Annual Science Report
 NASA Goddard Space Flight Center
Reporting | JUL 2007 – JUN 2008
NASA Goddard Space Flight Center
Reporting | JUL 2007 – JUN 2008
Organic and Inorganic Acids From Ion-Irradiated Ices
Project Summary
Scientists at the Cosmic Ice Laboratory with the Goddard Center for Astrobiology study the formation and stability of molecules under conditions found in outer space. During the past year, amino acids found in meteorites were investigated, including some acids not found in terrestrial biology. Carbonic acid, a very unstable molecule on Earth, also was studied since it will be stable under Martian conditions and environments in the outer solar system. These projects are part of the Comic Ice lab’s continuing contributions to understanding the chemistry of biologically-related molecules and chemical reactions in extraterrestrial environments.
Project Progress
In the Cosmic Ice Laboratory we simulate the low-pressure, low-temperature environments of outer space using a high-vacuum chamber and a cryostat. Ice samples are prepared on a pre-cooled mirror inside the cryostat and either irradiated with 1-MeV protons to simulate cosmic-ray bombardment, photolyzed to simulate vacuum-uv exposure, or irradiated with 10-keV electrons to simulate a variety of magnetospheric and circumstellar environments.
In this period, we focused on two projects: 1. studies of the amino acids isovaline and valine, and 2. the formation and stability of carboxylic acids relevant to Mars and satellites of the outer solar system. The motivation for the amino-acid study is that isovaline is a non-biological meteoritic amino acid that is impossible to make by the Strecker process. Moreover, past experiments with the low-temperature energetic processing of ices have uniformly failed to show isovaline formation. We have examined the synthesis, destruction, isomerization, and racemization of both isovaline and valine in our laboratory. Our second project during this period relates to ongoing and future Mars and outer solar system missions that will search for the evidence of surface organics. Our new work began with the study of the stability of several organic acids in low-temperature environments.
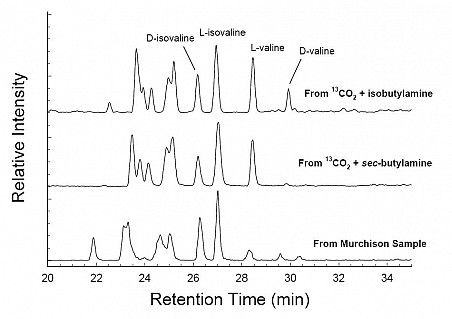
Our amino-acid experiments showed that isovaline’s enhancement over valine in the Murchison meteorite is not due to either a difference in thermal or radiation stabilities between the two molecules or to a radiation-induced isomerization that favors one molecule over the other. These results were obtained by chemical analyses of residual material from ion-irradiated isovaline and valine in the absence and presence of water-ice. As for these amino acids’ formation, our work shows unequivocal evidence, for the first time, of isovaline and valine syntheses by energetic processing of a cosmic-ice analog. In these experiments, irradiated 13CO2 + butylamine ices were warmed under vacuum and the residues were analyzed by LC/ToF-MS in Goddard’s Astrobiology Analytical Laboratory (see Report by Dr. Jason Dworkin). Protocols involved blanks and standards.
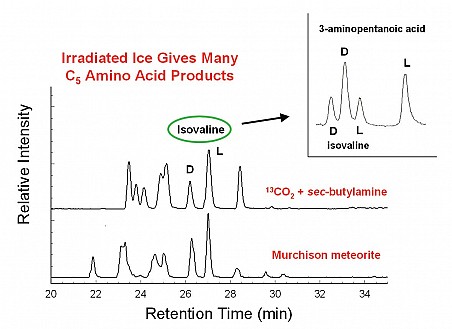
Although the reactants for our amino-acid formation experiments were either achiral (isobutylamine) or a racemic mixture (sec-butylamine), an enantiomeric excess (ee) of L over D for isovaline and valine appeared in our residues, as shown in Figure 1. One possible explanation for these strikingly large ee values is that another C5 amino acid isomer is co-eluting with L-isovaline and L-valine, artificially boosting each molecule’s chromatographic peak. GCA Co-Investigators Drs. Jason Dworkin and Danny Glavin examined most of the twelve different C5 amino-acid isomers and found that co-elution by 3-aminopentanoic acid explained the apparent L-excesses. Figure 2 shows that the D and L peaks identified with 3-aminopentanoic acid co-elute with the L-isovaline and L-valine, respectively.
New work related to the stability of acids on Mars and outer solar system surfaces has begun in the Cosmic Ice Laboratory. This year’s focus was on carbonic acid (H2CO3), which can be formed at low temperature by photolysis and radiolysis of H2O + CO2 ice mixtures. Carbonic acid’s thermal and radiation stability in, for example, the Martian regolith is a critical piece of physical-chemical data for understanding the chemistry of amino- and carbonic-acid mixtures. We formed carbonic acid by freeze-drying an irradiated H2O + CO2 ice. Once water was removed, the decrease of carbonic acid’s spectral bands at a specific temperature was used to calculate a vapor pressure for the compound. The slope of a ln(vapor pressure) vs 1/T graph then was used to calculate the heat of sublimation, ΔH = 66.3 kJ/mol, a value similar to what we have measured for two other carboxylic acids with the same technique. We also determined that carbonic acid’s proton radiation half-life was reached after a dose of 4-10 eV / molecule at 10 K.
Other activities, including outreach, from the past year
- Further work on low-temperature reactions of sulfur-containing molecules, motivated by possible connections to sulfur- and sulfate- reducing bacteria.
- Continued experiments with keV electron gun comparing effects of 1-MeV proton and 100-keV electron irradiations
- Proposed and organized three oral sessions and one poster session for the upcoming Astrobiology Science Conference (AbSciCon 2008) meeting in Santa Clara, California – The meeting’s organizers said that these sessions were in the top three, of thirty-six, in number of abstract submissions.
- Oral presentations at NASA workshop on icy satellites in the outer solar system held in Colorado in August, 2007 and a workshop on solar system ices held in California in May, 2008; oral presentation at April, 2008 AbSciCon meeting
- Two posters and one talk presented at the AAS Division for Planetary Sciences meeting in October, 2007 – one poster concerned the formation of meteoritic isovaline and one on stability of carboxylic acids
- Oral presentation at the IAU’s Bioastronomy 2007 meeting in Puerto Rico
- Manuscript submitted for publication for the IAU’s Bioastronomy 2007 meeting in Puerto Rico
- Chapter for the University of Arizona Press’s new book on trans-Neptunian objects (appeared spring, 2008), with an astrobiology section
- Manuscript scheduled for August publication in Astrobiology, on amino acids formed during the low-temperature irradiation of nitrile-containing molecules and molecular mixtures; authors include three GCA team members and one GCA intern
- About fifteen manuscripts reviewed for publication (Astrobiology, Astrophysical Journal, Icarus, Astronomy & Astrophysics, J. Phys. Chem., J. Chem. Education, Chemical Physics, and Chemical Physics Letters)
- Proposals reviewed for two NASA panels
- Assisted with Goddard Center for Astrobiology summer interns (one field trip to the NRAO in Green Bank, a laboratory tour, and a luncheon presentation)
- One college-level astrobiology course taught
-
PROJECT INVESTIGATORS:
-
RELATED OBJECTIVES:
Objective 2.2
Outer Solar System exploration
Objective 3.1
Sources of prebiotic materials and catalysts
Objective 7.1
Biosignatures to be sought in Solar System materials
