2015 Annual Science Report
 Massachusetts Institute of Technology
Reporting | JAN 2015 – DEC 2015
Massachusetts Institute of Technology
Reporting | JAN 2015 – DEC 2015
Taphonomy of Microbial Ecosystems
Project Summary
We perform experiments to understand shapes, molecules and isotopic signals of microbial processes in modern and old sediments. Experimental studies of microbial interactions with sediments, ions in the solution and the flow help us elucidate mechanisms that may have shaped sandy surfaces and preserved fossils on these surfaces at the dawn of animal life. Culture-based studies of isotopic fractionations produced by microbial processes and microbial membrane lipids help us interpret corresponding signals in the rock record and modern environments.
Project Progress
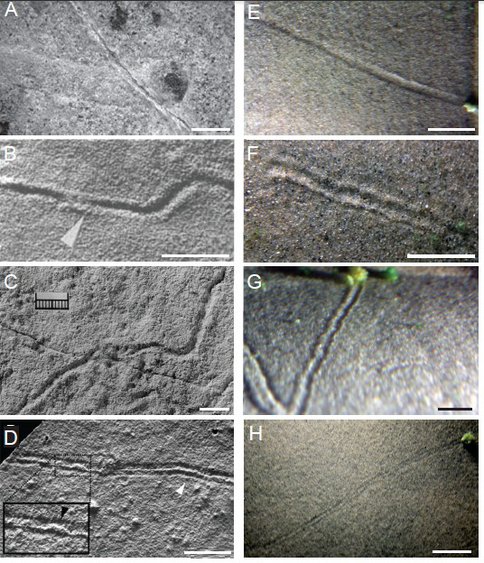
Dr. Giulio Mariotti, a former postdoctoral scholar, established an experimental system to study the biogeochemical properties of and microbial growth on sandy sediments in the presence of bedforms and oscillatory flow. Dr. Mariotti has demonstrated for the first time that interactions between oscillatory flow and cm-scale microbial aggregates that produce elongated trails on the surfaces of sand and silt beds. These findings provide an alternative, microbial mechanism for the formation of various surface trails which abounded in late Ediacaran and early Paleozoic sandstones and siltstones and were invariably attributed to early animals. An article describing these results is in press (Mariotti et al., JSR, in press). Dr. Mariotti is preparing another article describing the interactions between the oscillatory flow over bedforms and microbial growth. He is now an assistant professor at Louisiana State University.
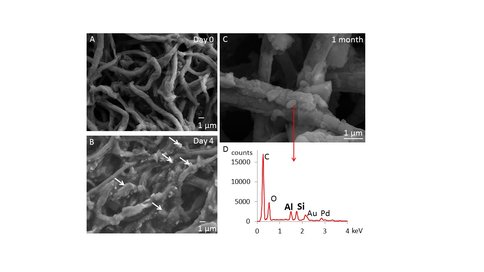
Sharon Newman, a PhD student, studies the preservation of microbes and tissues in siliciclastic environments. Using insights developed by Dr. Mariotti, Sharon experimentally investigated microbial fossilization under conditions which did not mobilize sand grains, but mobilized clay and fine silt. She has identified hydrodynamic and sedimentological conditions (fine sediment load and agitation) that resulted in the best preservation of cyanobacteria and tested the hypothesis about the ability of higher concentrations of silica in the past to promote the fossilization in sandstones and siltstones. The sheaths of thin filamentous cyanobacteria can trap suspended fine sediment and become encased in clay-rich, uniformly thick molds within days. In the absence of suspended and trapped clay nuclei, aluminosilicates precipitate sporadically around cyanobacterial filaments, but take more than a month to do so. High silica concentrations (greater than 0.1 mM) facilitate the precipitation of aluminosilicates in the absence of nuclei and also produce the smoothest and most continuous molds around cyanobacterial filaments when clay nuclei are present. These experiments show that the preservation of filamentous photosynthetic organisms and millimeter-scale microbial textures can occur under fully oxic conditions in sandy and silty environments, if abundant suspended clays and fine silt and high silica concentrations are present. This mechanism can account for the preservation of similar features during the Ediacaran, with implications for the preservation of organic matter in sandstones, siltstones and shales. Sharon presented these results at AbSciCon, Goldschmidt and GSA conferences in 2015 and received the GSA Geobiology and Geomicrobiology Division Award for the best student oral presentation. Sharon submitted the first manuscript describing her results (Newman et al., in review) and is preparing another one. Matt Joss, a former MSc student supervised by Drs. Bosak and Rothman experimentally investigated the preservation of cyanobacterial aggregates by the precipitation of authigenic minerals as a function of iron availability and the activity of heterotrophic sedimentary microbes. He developed an experimental system and found that 100 μm to 1 mm-wide spherical aggregates of the cyanobacterium Spirullina did not retain their spherical shape during early diagenesis even when authigenic minerals formed in close association with the organic matter. These experiments argue against interpretations of some mineral- and organic-rich ovoid and spherical structures in Cryogenian carbonates as fossilized remnants of microbial mats. Matt submitted his thesis (Joss, 2015) and obtained a Masters degree.
Bosak collaborated with Dr. Dan Lahr, a biologist at University of Sao Paulo on the evolution of silicifying testate amoebae (Lahr et al., 2015). Bosak and Emily Matys, an MIT graduate student also collaborated with Dr. David McNeil at the Geological Survey of Canada on the preservation of agglutinated foraminifera (McNeil et al., 2015). Kelsey Moore worked on her undergraduate thesis with Dr. Sara Pruss at Smith College, graduated in 2015 and joined the Bosak Lab. Mentored by Dr. Pruss, Kelsey found new assemblages of agglutinated fossils in the Cryogenian cap carbonates of Mongolia and Zambia and new localities of the Rasthof Formation, Namibia. She also found assemblages of coiled, pyritized molds of Obruchevella in the Cryogenian cap carbonates from Arctic Alaska. Bosak supervised Kelsey’s mineralogical and compositional characterizations and interpretations of these structures. Kelsey presented her work at the Fall GSA Meeting and is preparing two manuscripts describing this work. Dr. Phoebe Cohen, a former MIT postdoc and now an assistant professor at Williams College published an article describing organic fossils with possible red algal affinities (Cohen et al., 2015). These fossils were preserved in carbonate strata of the Cryogenian interglacial interval in Mongolia.
Dr. Xiaolei Liu, a postdoctoral scholar advised by Drs. Summons and Bosak has identified novel archeal glycerol dialkyl glycerol tetraethers with a cyclohexyl moiety in Green Lake, NY and has noted the occurrence of these lipids in sulfidic environments. He has submitted a manuscript describing this work and its implications for modern and ancient sediments deposited in sulfidic conditions (Liu et al., in review). Bosak collaborated with Dr. Shuhei Ono (MIT), Orit Sivan (BGU, Israel) and Sasha Turchyn (Cambridge U) on studies of microbial sulfate reduction. One manuscript was published (Ono et al., 2015), one was submitted (Antler et al., submitted) and one, led by Dr. Shikma Zaarur, a former postdoctoral scholar, is in preparation.
Dr. Shane O’Reilly, a postdoctoral scholar advised by Drs. Summons and Bosak has investigated the formation of modern ooids in Pigeon Cay, Bahamas, as possible records of seawater chemistry and microbial processes. Current abiotic biological models of ooid formation do not explain the large abundance of organic matter in modern ooids, whereas microbial models of ooid formation do not account for hydrodynamic conditions in areas where ooids are typically found. Shane identified photosynthetic eukaryotes and Alphaproteobacteria as principal sources of fresh organic matter in ooids, but found a strong signal of anaerobic heterotrophic alteration and mineralization of organic matter in the pool of bound lipids in ooid carbonate. This indicates that the organic matter bound within ooids in Pigeon Cay record primarily the degradation of organic matter by sulfate-reducing bacteria and other anaerobes. Shane’s measurements suggest that some ooid carbonate forms within the microbially-colonized areas of unmobilized sediment, in contrast to the abiotic models of ooid formation which assume that ooid carbonate precipitates only in the surf zone and while the ooids are mobilized. Shane recently submitted a manuscript describing his findings (O’Reilly et al., submitted).
Dr. Florence Schubotz, a former postdoctoral scholar supervised by Drs. Summons and Bosak sought to identify possible sources of abundant phosphate-lacking intact polar lipids that occur in some anaerobic marine sediments, oxygen minimum zones and other anoxic or suboxic areas of oceans. Flo used five different phosphate-limited sulfate reducing bacteria as model organisms and analyzed the composition of lipid membranes in phosphate-replete and phosphate-limited media. She found that phosphate-lacking lipids replaced from 80-100% of phospholipids in the membranes of all tested sulfate reducers during phosphate limitation. All phosphate-limited sulfate reducers synthesized glycolipids and some also synthesized betaine lipids and ornithine lipids. Flo’s findings reveal sulfate reducing bacteria as possible sources of betaine lipids and glycolipids in some anaerobic environments and suggest that phosphate limitation may affect microbes even in environments where phosphate was not considered to be a limiting nutrient. Flo is currently preparing manuscripts to describe her findings.
References:
Antler, G., Turchyn, A.V.T., Ono, S., Sivan, O. and Bosak, T. Combined 34S, 33S and 18O isotope fractionations record different intracellular steps of microbial sulfate reduction, submitted.
Cohen, P.A., Macdonald, F.A., Pruss, S.B., Matys, E.D., Bosak, T. 2015. Fossils of putative marine algae from the Cryogenian glacial interlude of Mongolia. Palaios, 30, 238-247.
Lahr, D.J.G., Bosak, T., Lara, E., Mitchell, E.A.D., Phanerozoic diversification of silica-cycling testate amoebae and the strengthened coupling of terrestrial C and Si cycles, PeerJ 3:e1234 https://dx.doi.org/10.7717/peerj.1234
Liu, X., De Santiago Torio, A., Bosak, T., Summons, R.E. Novel archaeal tetraether lipids with cyclohexyl rings identified in Green Lake, NY and other sulfidic lacustrine settings, submitted.
Mariotti, G., Pruss, S.B., Ai, X. and Bosak, T. Microbial origin of early animal trace fossils?, JSR, in press.
McNeil, D., Shulze, H.G., Matys, E.D., and Bosak, T. 2015. Raman spectroscopic analysis of carbonaceous matter and silica in the test walls of recent and fossil agglutinated foraminifera, AAPG Bulletin, 99, 1081-1097.
Newman, S.A., Mariotti, G., Pruss, S.B., Bosak, T. A recipe for cyanobacterial fossilization in siliclastic environments, submitted.
Ono, S., Sim, M.S., Bosak, T. Predictive isotope model connects microbes in culture and nature, PNAS, 111, 18102-18103.
O’Reilly, S.S., Mariotti, G., Winter, A.R., Newman, S.A., Matys, E.D., McDermott, F.,Pruss, S.B., Summons, R.E., Klepac-Ceraj, V., Bosak, T. Microbial diversity and molecular biosignatures associated with surf zone and subtidal ooids at Pigeon Cay, the Bahamas, submitted.
Publications
-
Cohen, P. A., Macdonald, F. A., Pruss, S., Matys, E., & Bosak, T. (2015). FOSSILS OF PUTATIVE MARINE ALGAE FROM THE CRYOGENIAN GLACIAL INTERLUDE OF MONGOLIA. PALAIOS, 30(3), 238–247. doi:10.2110/palo.2014.069
-
Lahr, D. J. G., Bosak, T., Lara, E., & Mitchell, E. A. D. (2015). The Phanerozoic diversification of silica-cycling testate amoebae and its possible links to changes in terrestrial ecosystems. PeerJ, 3, e1234. doi:10.7717/peerj.1234
-
McNeil, D. H., Schulze, H. G., Matys, E., & Bosak, T. (2015). Raman spectroscopic analysis of carbonaceous matter and silica in the test walls of recent and fossil agglutinated foraminifera. AAPG Bulletin, 99(06), 1081–1097. doi:10.1306/12191414093
-
Ono, S., Sim, M. S., & Bosak, T. (2014). Predictive isotope model connects microbes in culture and nature. Proceedings of the National Academy of Sciences, 111(51), 18102–18103. doi:10.1073/pnas.1420670111
-
Mariotti, G., Pruss, S. B., Ai, X., Perron, J. T., & Bosak, T. (2016). Microbial Origin of Early Animal Trace Fossils?. Journal of Sedimentary Research, 86(4), 287–293. doi:10.2110/jsr.2016.19
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Phoebe Cohen
Co-Investigator
Francis Macdonald
Co-Investigator
Giulio Mariotti
Co-Investigator
Shuhei Ono
Co-Investigator
Sara Pruss
Co-Investigator
Dan Rothman
Co-Investigator
Matt Joss
Collaborator
Vanja Klepac-Ceraj
Collaborator
Daniel Lahr
Collaborator
Matthew Marcus
Collaborator
Emily Matys
Collaborator
David McNei
Collaborator
Kelsey Moore
Collaborator
Sharon Newman
Collaborator
Taylor Perron
Collaborator
Florence Schubotz
Collaborator
Georg Shulze
Collaborator
Nobumichi Tamura
Collaborator
Shikma Zaarur
Collaborator
-
RELATED OBJECTIVES:
Objective 2.1
Mars exploration.
Objective 4.1
Earth's early biosphere.
Objective 4.2
Production of complex life.
Objective 5.1
Environment-dependent, molecular evolution in microorganisms
Objective 5.2
Co-evolution of microbial communities
Objective 6.1
Effects of environmental changes on microbial ecosystems
Objective 7.1
Biosignatures to be sought in Solar System materials
Objective 7.2
Biosignatures to be sought in nearby planetary systems