2013 Annual Science Report
 NASA Jet Propulsion Laboratory - Icy Worlds
Reporting | SEP 2012 – AUG 2013
NASA Jet Propulsion Laboratory - Icy Worlds
Reporting | SEP 2012 – AUG 2013
Investigation 2: Survivability on Icy Worlds
Project Summary
Investigation 2 focuses on survivability. As part of our survivability investigation, we examine the similarities and differences between the abiotic chemistry of planetary ices irradiated with ultraviolet photons (UV), electrons, and ions, and the chemistry of biomolecules exposed to similar conditions. Can the chemical products resulting from these two scenarios be distinguished? Can viable microbes persist after exposure to such conditions? These are motivating questions for our investigation.
Project Progress
The following Co-Is and Collaborators have been conducting research as summarized below. Johnson, Hodyss and Ponce (JPL), carried out spectroscopy and viability of spores in ice; Strazzulla (INAF, Italy), investigated sulfur containing ices under the influence of ion induced radiation processing.
Co–Investigators Paul Johnson, Robert Hodyss, Aaron Noell and Adrian Ponce, along with JPL summer intern Halley Darrach continued an investigation into the spectral properties of bacterial spores in cryogenic ices as well as their viability under solar radiation. Bacterial spores are one of the toughest and most durable forms of life on earth. Can bacterial spores embedded in near surface Europan ice survive the Jovian radiation environment? Can either the spores or their organic radiolysis products be detected spectroscopically through remote sensing?
Bacillus subtillus was chosen for not only its ability to readily generate spores, but its well-established use as a model species. It is a useful tracker of UV irradiation effects because of its high resilience to a variety of extreme conditions. As such it is an important indicator species for forward contamination in the context of planetary protection. Purified spores were deposited onto silicon-oxide coated aluminum mirrors in both a monolayer, and multilayer form. The multilayer form, which generated a substantially increased FT-IR signal, was initially used to more clearly identify the known absorptive features of spores at standard temperature. The monolayer was then substituted in to make the radiation effects more reproducible. The monolayer spores were stamped onto half of the mirror (the remaining half used as a background blank), whereas the multilayer took the form of a noticeable spore spot on the mirror ~2mm in diameter. Once prepared, the mirrors were mounted on a cryostat and transferred to a vacuum apparatus for UV irradiation and FT-IR analysis. For viability studies the spores were deposited on nickel plated stainless steel sample tabs. Here monolayers were used to allow for more accurate post-UV viability testing ensuring that all spores received an equal UV dosage and were not protecting one another.
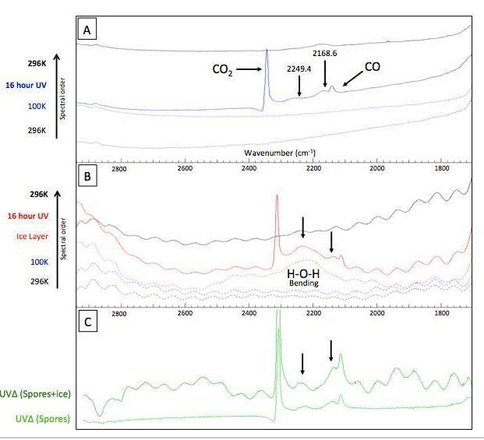
Briefly, we found that B. subtilis spores generate low molecular weight photoproducts when irradiated with UV at low temperature and pressures (100K and 10-9 Torr). After irradiation, absorption bands were observed at 2249.4 and 2168.7 cm-1, which is within the general region of nitrile containing groups, though no positive identification has yet been achieved (Figure 2-1). Looking at the evolved gases with a mass spectrometer while heating the samples showed that the associated irradiation-induced photoproducts generate secondary products upon the ionization with masses of 14.2 and 40.2 amu. The addition of a thin (0.25μm) water-ice layer on top of the spores did not appear to alter or interfere with the UV induced photochemistry and accompanying products.
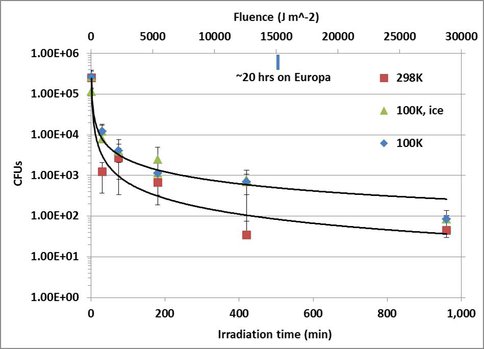
Results of the viability work are seen in figure 2-2. We find increased viability of spores irradiated at 100K vs room temperature. Future research will greatly expand the scope of the project to include (i) spore survivability as a function of ice thickness, (iii) spore spectral signature alteration as a function of ice thickness, and (iv) the identification of the aforementioned photoproducts with accompanying chemistry.
Collaborator G. Strazzulla of the INAF-Catania Astrophysical Observatory group in collaboration with the group at CIMAP-GANIL in Caen (France) performed experiments on effects induced by fast ions colliding with solids of astrophysical interest. The project is aimed at measuring of formation yields of specific molecules after implantation of reactive (H, C, N, O, S etc), multiply charged ions at different energies in water ice with relevant application to the chemistry of the icy moons in the outer solar system. Ion beams have been obtained at GANIL after a selective call for proposals to which we successfully applied.
The results obtained in the last year are relative to implantation of 100’s keV Sn+ (n =7, 9, 11) into thick films (i.e. thicker than the penetration depth of the ions) of water ice, The results of our experiments indicate that implantation produces hydrated sulfuric acid with yields that increase with ion energy, from 0.12 molecules /ion for 35 keV ions to 0.64 molecules/ ion for 200 keV ions. We have also searched for the production of SO2 and H2S, but we were not able to find any evidence for their synthesis. We conclude that sulfur ion implantation is the dominant formation mechanism of hydrated sulfuric acid at Europa. The suggestion that the observed distribution of sulfuric acid on the surface is well correlated with the local flux of sulfur ions find a full explanation by present experimental data and indicate that sulfuric acid was produced after implantation with different charge states at different temperatures.
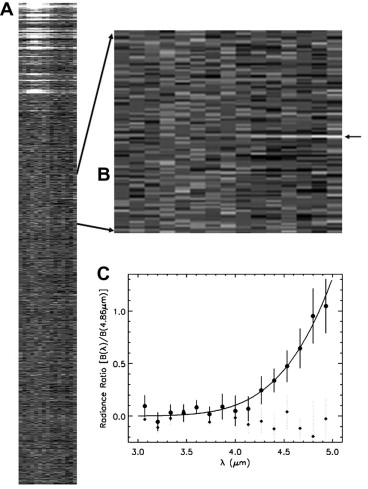
Co-I C. Sotin and a team of investigators analyzed a sequence of high spatial resolution near-infrared spectra acquired with VIMS at 0.025 s intervals during a 74 km altitude flyover of the South Pole of Enceladus by the Cassini spacecraft on 14 April 2012 UTC. A thermal-emission spectrum covering 3- to 5-μm wavelengths was detected as the field of view crossed one of the four major fissures, Baghdad Sulcus, within 1 km of 82.36S latitude and 28.24W longitude. This spectrum was interpreted as thermal emission from a linear fissure with temperature 197 ± 20 K and width 9 m. At the above wavelengths, the spectrum is dominated by the warmest temperature component. Looking downward into the fissure at only 13° from the vertical, it was concluded that our results measured the temperature of the interior fissure walls (and the H2O vapor) at depths within 40 m of the surface.
As a part Investigation 2, Co-I Murthy Gudipati reported the results of a laboratory investigation conducting earlier employing a two-color laser-desorption laser-ionization time-of-flight mass spectroscopic method (2C-MALDI-TOF), similar to matrix-assisted laser desorption and ionization time-of-flight (MALDI-TOF) mass spectroscopy. Laboratory results with polycyclic aromatic hydrocarbon (PAH) probe molecules embedded in water-ice at 5 K show for the first time that hydrogenation and oxygenation are the primary chemical reactions that occur in astrophysical ice analogs when subjected to Lyα radiation. They found that hydrogenation can occur over several unsaturated bonds and the product distribution corresponds to their stabilities. Multiple hydrogenation efficiency is found to be higher at higher temperatures (100 K) compared to 5 K—close to the interstellar ice temperatures. Hydroxylation is shown to have similar efficiencies at 5 K or 100 K, indicating that addition of O atoms or OH radicals to pre-ionized PAHs is a barrierless process. These studies—the first glimpses into interstellar ice chemistry through analog studies—show that once accreted onto ice grains PAHs lose their PAH spectroscopic signatures through radiation chemistry, which could be one of the reason for the lack of PAH detection in interstellar ice grains, particularly the outer regions of cold, dense clouds or the upper molecular layers of protoplanetary disks.
Publications
-
Allodi, M. A., Baragiola, R. A., Baratta, G. A., Barucci, M. A., Blake, G. A., Boduch, P., … Strazzulla, G. (2013). Complementary and Emerging Techniques for Astrophysical Ices Processed in the Laboratory. Space Sci Rev, 180(1-4), 101–175. doi:10.1007/s11214-013-0020-8
-
Boduch, P., Domaracka, A., Fulvio, D., Langlinay, T., Lv, X. Y., Palumbo, M. E., … Strazzulla, G. (2012). Chemistry induced by energetic ions in water ice mixed with molecular nitrogen and oxygen. A&A, 544, A30. doi:10.1051/0004-6361/201219365
-
Cassidy, T. A., Paranicas, C. P., Shirley, J. H., Dalton III, J. B., Teolis, B. D., Johnson, R. E., … Hendrix, A. R. (2013). Magnetospheric ion sputtering and water ice grain size at Europa. Planetary and Space Science, 77, 64–73. doi:10.1016/j.pss.2012.07.008
-
Dalton, J. B., & Pitman, K. M. (2012). Low temperature optical constants of some hydrated sulfates relevant to planetary surfaces. Journal of Geophysical Research: Planets, 117(E9), n/a–n/a. doi:10.1029/2011je004036
-
Dalton, J. B., Cassidy, T., Paranicas, C., Shirley, J. H., Prockter, L. M., & Kamp, L. W. (2013). Exogenic controls on sulfuric acid hydrate production at the surface of Europa. Planetary and Space Science, 77, 45–63. doi:10.1016/j.pss.2012.05.013
-
Ding, J. J., Boduch, P., Domaracka, A., Guillous, S., Langlinay, T., Lv, X. Y., … Strazzulla, G. (2013). Implantation of multiply charged sulfur ions in water ice. Icarus, 226(1), 860–864. doi:10.1016/j.icarus.2013.07.002
-
Goguen, J. D., Buratti, B. J., Brown, R. H., Clark, R. N., Nicholson, P. D., Hedman, M. M., … Blackburn, D. G. (2013). The temperature and width of an active fissure on Enceladus measured with Cassini VIMS during the 14 April 2012 South Pole flyover. Icarus, 226(1), 1128–1137. doi:10.1016/j.icarus.2013.07.012
-
Lv, X. Y., De Barros, A. L. F., Boduch, P., Bordalo, V., Da Silveira, E. F., Domaracka, A., … Strazzulla, G. (2012). Implantation of multiply charged carbon ions in water ice. A&A, 546, A81. doi:10.1051/0004-6361/201219886
-
Strazzulla, G. (2013). Crystalline and amorphous structure of astrophysical ices. Low Temp. Phys., 39(5), 430. doi:10.1063/1.4807045
- Ding, J.J., Boduch, P., Domaracka, A., Langlinay, T., Lv, X.Y., Palumbo, M.E., Rothard, H. & Strazzulla, G. (2012). Implantation of Sn+ ions on the Galilean moons. EPSC.
- Noell, A.C., Ely, T., Bolser, D., Darrach, H., Hodyss, R., Johnson, P.V. & Ponce, A. (2014). Spectroscopy and viability of Bacillus subtilis spores after ultraviolet irradiation: Implications for the detection of potential bacterial life on Europa. Astrobiology.
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Paul Cooper
Co-Investigator
Brad Dalton
Co-Investigator
Kevin Hand
Co-Investigator
Robert Hodyss
Co-Investigator
Paul Johnson
Co-Investigator
Adrian Ponce
Co-Investigator
Christophe Sotin
Co-Investigator
Louis Allamandola
Collaborator
Bob Carlson
Collaborator
Giovanni Strazzulla
Collaborator
-
RELATED OBJECTIVES:
Objective 1.1
Formation and evolution of habitable planets.
Objective 2.2
Outer Solar System exploration
Objective 3.2
Origins and evolution of functional biomolecules
Objective 5.1
Environment-dependent, molecular evolution in microorganisms
Objective 5.3
Biochemical adaptation to extreme environments
Objective 6.1
Effects of environmental changes on microbial ecosystems
Objective 6.2
Adaptation and evolution of life beyond Earth