2012 Annual Science Report
 University of Wisconsin
Reporting | SEP 2011 – AUG 2012
University of Wisconsin
Reporting | SEP 2011 – AUG 2012
Project 3D: Constraints on Oxygen Contents in Earth’s Early Atmosphere and Implications for Evolution of Photosynthesis
Project Summary
The oxidation state of the atmosphere and oceans on the early Earth remains controversial. Although it is accepted by many workers that the Archean atmosphere and ocean were anoxic, hematite in the 3.46 billion-year-old (Ga) Marble Bar Chert (MBC) from Pilbara Craton, NW Australia has figured prominently in arguments that the Paleoarchean atmosphere and ocean was fully oxygenated. In this study, we report the Fe isotope compositions and U concentrations of the MBC, and show that the samples have extreme heavy Fe isotope enrichment. Collectively, the Fe and U data indicate a reduced, Fe(II)-rich, U-poor environment in the Archean oceans at 3.46 billion years ago. Given the evidence for photosynthetic communities provided by broadly coeval stromatolites, these results suggests that an important photosynthetic pathway in the Paleoarchean oceans may have been anoxygenic photosynthetic Fe(II) oxidation.
Project Progress
The evolution of photosynthesis was a critical evolutionary step in the history of life on Earth, and it has been proposed that anoxygenic photosynthesis preceded oxygenic photosynthesis, largely on the basis that the Archean Earth atmosphere and oceans are generally considered to have been anoxic. The first rise in atmospheric oxygen, termed the “Great Oxidation Event” (GOE), has been proposed to have occurred between ~2.45 and 2.2 Ga. The GOE model was developed based on geologic occurrences of detrital pyrite, siderite, and uraninite in sediments deposited before the GOE, as well as retention of Fe in paleosols after the GOE. Support for the GOE model comes from the disappearance of mass-independent fractionation of S isotope after the GOE. Recent geochemical studies, however, increasingly provide evidence for a more complex evolution of atmospheric O2 levels prior to the GOE as compared to a simple a “step-function”.
In this study, we focus on the origin and environmental significance of hematite from the 3.46 Ga Marble Bar Chert (MBC) from the Pilbara craton, Western Australia. Other workers have interpreted hematite in the MBC to have precipitated from a fully-oxygenated Archean ocean at the time of deposition at 3.46 Ga, which in turn would suggest that oxygenic photosynthesis had evolved prior to that time. We combine Fe isotopes, which can constrain the extent of oxidation, with U-Th-Pb isotopes, which provide an estimate of seawater U contents and tests for post-depositional alteration. Our results indicate that hematite in the MBC was precipitated by a very small extent of oxidation from an Fe(II)-rich, U-poor ocean at 3.46 Ga. We conclude, therefore, that hematite in the 3.46 Ga MBC cannot be used to infer oxygenic photosynthesis prior to 3.5 Ga.
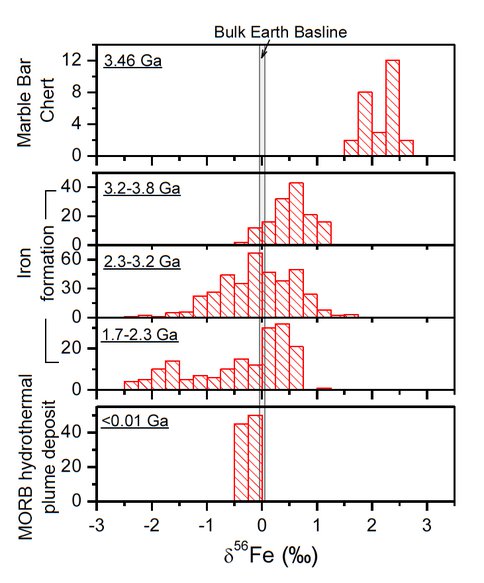
The MBC samples have very high δ56Fe values, ranging between +1.53 ‰ and +2.63 ‰, that define the upper limit of measured δ56Fe values for terrestrial samples. The likely source of aqueous Fe(II) that was oxidized to form hematite in the MBC was hydrothermal fluids, which should have had a δ56Fe value of around 0 ‰, or slightly negative. Oxidation of aqueous Fe(II) in modern marine hydrothermal systems produces precipitates that have slightly negative δ56Fe values, reflecting essentially quantitative oxidation. The very high δ56Fe values of hematite in the MBC, therefore, do not support a fully oxidized Archean ocean. Dispersion/reaction modeling indicates that very low oxidation rates and low degrees of Fe(II) oxidation are needed to produce the highly positive δ56Fe values. In particular, modeling strongly suggests that the photic zone of the 3.46 Ga ocean was reduced and buffered by excess Fe(II). In fact, modeling suggests that photic zone oxygen contents were less than 10-3 mmicromolar , less than 0.0003% of modern photic zone levels.
Figure 1. Comparison of Fe isotope compositions of the MBC samples from the ABDP-1
drill core (this study), Archean and Proterozoic iron formation (compiled from literature),
and modern (<16k yrs) hydrothermal plume precipitates from mid-ocean ridge environments (also from the literature).
Publications
-
Li, W., Czaja, A. D., Van Kranendonk, M. J., Beard, B. L., Roden, E. E., & Johnson, C. M. (2013). An anoxic, Fe(II)-rich, U-poor ocean 3.46 billion years ago. Geochimica et Cosmochimica Acta, 120, 65–79. doi:10.1016/j.gca.2013.06.033
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Francois-Xavier A'bazac
Postdoc
Andrew Czaja
Postdoc
Weiqiang Li
Postdoc
-
RELATED OBJECTIVES:
Objective 4.1
Earth's early biosphere.
Objective 6.1
Effects of environmental changes on microbial ecosystems
Objective 7.1
Biosignatures to be sought in Solar System materials
Objective 7.2
Biosignatures to be sought in nearby planetary systems