2012 Annual Science Report
 NASA Jet Propulsion Laboratory - Titan
Reporting | SEP 2011 – AUG 2012
NASA Jet Propulsion Laboratory - Titan
Reporting | SEP 2011 – AUG 2012
Task 3.1.2 Chemistry Active in Titan Dunes
Project Summary
The goal of this project is to demonstrate that chemical reactions can occur between atmospheric organics and water ice, even at the low temperature (ca. 100 K) of Titan’s surface, leading to the incorporation of oxygen to form molecules of astrobiological significance. Studies using a low temperature fluidized bed reactor demonstrate that amino acids, glycine specifically, are produced by tribochemical reactions driven by Aeolian processes in Titan’s dunes.
Project Progress
Co-Investigator Jack Beauchamp and graduate student Daniel Thomas have examined the efficiency of tribochemical reactions between complex organics and water ice as one option for chemistry in the dunes of Titan. Aeolian (wind driven) processes lead to tribochemical charging of the airborne organic particles that comprise Titan’s vast dune fields. Discharges between differentially charged particles leads to oxygen incorporation (from water ice) into complex CHN organics formed by photochemical processes in Titan’s atmosphere. Continued experiments with low temperature fluidized bed reactors have provided clear evidence for the formation of amides and amino acids, specifically glycine, starting from aminoacetonitrile, which has been produced in abundance in simulations of Titan’s atmospheric chemistry. Figure 1 shows the mass spectrum of the organics extracted from the reactor following extensive agitation with flowing cooled nitrogen. Collisional activation of the species observed at m/z 76 yields fragments identical to those observed with a sample of pure glycine, providing support for the assigned structure. These experiments provide clear evidence for the role of tribochemical reactions in Titan’s dunes leading to the formation of astrobiologically significant molecules, even at the low temperatures of Titan’s surface.
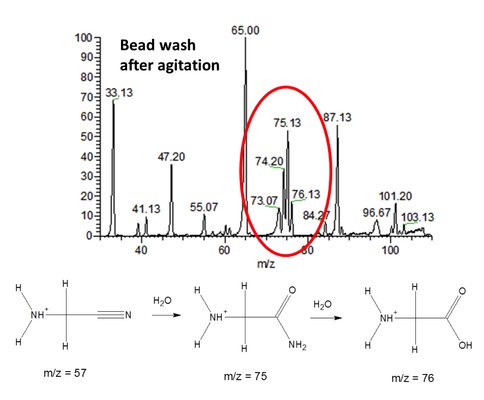 Figure 1. Mass spectrum showing organics extracted from fluidized bed reactor. Water ice reacts with aminoacetonitrile to form acetamide and glycine (observed as protonated species at m/z 75 and 76).
Figure 1. Mass spectrum showing organics extracted from fluidized bed reactor. Water ice reacts with aminoacetonitrile to form acetamide and glycine (observed as protonated species at m/z 75 and 76).
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Daniel Thomas
Graduate Student
-
RELATED OBJECTIVES:
Objective 2.2
Outer Solar System exploration
Objective 3.1
Sources of prebiotic materials and catalysts
