2012 Annual Science Report
 NASA Goddard Space Flight Center
Reporting | SEP 2011 – AUG 2012
NASA Goddard Space Flight Center
Reporting | SEP 2011 – AUG 2012
Fischer-Tropsch Type (FTT) Reactions in the Solar Nebula
Project Summary
We have pursued this research topic on two fronts over the past year. First we have almost completed construction of the new 10-station FTT experimental system, financed by NASA’s Exobiology R&A Program. This system replicates our original experimental setup in a much more compact fashion and should allow us sufficient flexibility to carry out several very long-term, low-temperature experiments while simultaneously having the capability to do a number of much faster experiments at higher temperatures. The second research front we have pursued is in trying to predict the “global” consequences of potentially extensive FTT reactions on the chemistry of the solar nebula and on protostars in general. In addition to the potential for FTT processes to produce interesting pre-biotic organic molecules such as methyl cyanide, various amino acids and more complex aromatic compounds, or the possibility that the macromolecular organic coating produced via FTT reactions could trap noble gases and carry these materials into planetary bodies, we believe that FTT reactions could have several additional, broader consequences for nebular chemistry – and even possibly for the chemical evolution of the galaxy.
Project Progress
We have pursued this research topic on two fronts over the past year. First we have almost completed construction of the new 10-station FTT experimental system, financed by NASA’s Exobiology R&A Program. This system replicates our original experimental setup in a much more compact fashion and should allow us sufficient flexibility to carry out several very long-term, low-temperature experiments while simultaneously having the capability to do a number of much faster experiments at higher temperatures. These faster experiments are typically run at 775 – 875K for from several days to several weeks. The lower temperature experiments, carried out at temperatures between 475 – 675K could require as much as 6 months to finish a single run, while the entire experiment could require as many as 20 sequential runs with the same catalyst – or at least 10 years to complete a single experiment.
Figure 1 – Schematic diagram of a single FTT system used to form organic compounds from a starting gas mixture of (typically) 550 torr of hydrogen, 25 torr of nitrogen, and 25 torr of a noble gas mixture that is 49% neon, 49% argon, 1% krypton and 1% xenon. The purpose of the experiments is to form macromolecular organic coatings that can be compared to the IOM found in meteorites and simultaneously trap noble gases for comparison with the planetary noble gas component found in primitive meteorites.
For each experiment we use on the order of 25cc of catalytic dust grains, flowing the gas mixture through the grains in a “fluidized bed” reaction scheme. The catalysts have ranged from pure iron particles, through various iron oxide, carbonate or hydroxide minerals, to crystalline silicates and even graphite. However, the majority of the experiments have been carried out using an amorphous iron silicate smoke synthesized in our laboratory in a hydrogen-oxygen flame containing trace quantities of silane and iron pentacarbonyl. Amorphous silica and magnesium silicate have been used as well, and all of these materials become much more effective catalysts as the reaction proceeds and as an organic coating develops on their surfaces. This growing macromolecular organic coating could be the long sought after “Phase Q” responsible for trapping the “Planetary” noble gases. Preliminary experiments at 875K demonstrated trapping of Xe and Kr: the lower temperature (and more realistic) studies will test our hypothesis that Phase Q really is the macromolecular coating that we produce in our runs.
The second research front we have pursued is in trying to predict the “global” consequences of potentially extensive FTT reactions on the chemistry of the solar nebula and on protostars in general. In addition to the potential for FTT processes to produce interesting pre-biotic organic molecules such as methyl cyanide, various amino acids and more complex aromatic compounds, or the possibility that the macromolecular organic coating produced via FTT reactions could trap noble gases and carry these materials into planetary bodies, we believe that FTT reactions could have several additional, broader consequences for nebular chemistry – and even possibly for the chemical evolution of the galaxy.
The remarkable discovery that oxygen isotopes in primitive meteorites had δ17O vs. δ18O that plotted along a line of slope 1 rather than along the typical slope 0.52 equilibrium fractionation line was published almost 40 years ago (Clayton, Grossman and Mayeda, 1973). However, a satisfactory, quantitative explanation for this observation has yet to be found, though many different explanations have been proposed. The original problem was not only to find a process to distribute oxygen isotopes along a slope 1 line, but also to find a mechanism that simultaneously increased the 16O content of the dust. This implicitly assumed that the oxygen isotopic composition of the Earth, Mars and most meteorites was close to the composition of the dust in the protosolar nebula. However, the oxygen isotopic composition of the sun, as measured by the Genesis Mission (McKeegan, et al., 2011) is considerably enriched in 16O relative to the composition of terrestrial oxygen as exemplified in SMOW. A photochemical approach to understanding the slope 1 distribution in solar system solids as well as the difference in isotopic composition between the sun and meteorites, Mars and the Earth has been pursued by several groups (Clayton, 2002; Lyons and Young, 2005; Yurimoto and Kuramoto, 2004).
Photochemical Self-Shielding models come in at least three distinct varieties with different degrees of commonality. All rely on the exhaustion of 12C16O dissociative photons as the radiation source penetrates through the gas and this fractionation can be calculated precisely for each dissociative band of CO. In particular the model assumes that most of the 16O-depleted oxygen combines with abundant hydrogen to produce water or ice and that the CO in these regions would be significantly enriched in C16O that, due to the presumed stability of the CO molecule, then serves as a permanent trap for 16O. FTT reactions convert CO (plus hydrogen) into hydrocarbons and water, thus eliminating the long-term stability of the CO molecule and putting the “trapped” 16O back in circulation. If FTT reactions operated extensively in the Solar Nebula, the net effect of any Photochemical Self Shielding reactions would be quickly eliminated as the water from the FTT reactions mixed with the isotopically enriched water produced by self-shielding.
In addition to their effects on nebular chemistry, we are just beginning to realize that FTT reactions could have a much larger effect on chemical speciation in the galaxy: specifically, on the structure and composition of carbonaceous dust. More than 95% of silicate minerals and other oxides found in meteorites were melted, or vaporized and recondensed in the Solar Nebula prior to their incorporation into meteorite parent bodies (Boynton, 1985). Gravitational accretion energy and heating via radioactive decay further transformed oxide minerals accreted into planetesimals. In such an oxygen-rich environment the carbonaceous dust that fell into the nebula as an intimate mixture with oxide grains should have been almost completely converted to CO. While some pre-collapse, molecular-cloud carbonaceous dust does survive (Bernatowicz and Cowisk, 1997), much in the same manner as do pre-solar oxide grains (Nittler et al. 1997), such materials constitute only a few percent of meteoritic carbon and are clearly distinguished by elevated D/H, 15N/14N, 13C/12C ratios or noble gas patterns.
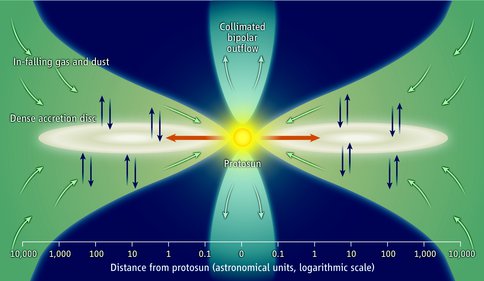
We argue that nearly all of the carbon in meteorites was synthesized in the Solar Nebula from CO and that this CO was generated by the reaction of carbonaceous dust with solid oxides, water or OH. It is probable that some fraction of carbonaceous dust that is newly synthesized in the Solar Nebula is also converted back into CO by additional thermal processing. CO processing might occur on grains in the outer nebula through irradiation of CO-containing ice coatings (Ciesla and Sandford, 2012) or in the inner nebula via Fischer-Tropsch type (FTT) reactions on grain surfaces (Nuth et al. 2008). Large-scale transport of both gaseous reaction products and dust from the inner nebula out to regions where comets formed would spread newly formed carbonaceous materials throughout the solar nebula (Nuth et al. 2000).
Figure 2 – Large-scale circulation in the solar nebula mixes materials outward after processing in the higher pressure and temperature regions of the inner nebula
Carbon dust in the ISM might easily be described as inorganic graphite or amorphous carbon, with relatively low structural abundances of H, N, O and S (Jones and Nuth, 2011). Products of FTT reactions or organics produced via irradiation of icy grains contain abundant aromatic and aliphatic hydrocarbons, aldehydes, keytones, acids, amines and amides (Hill and Nuth, 2003). The net result of the massive nebular carbon cycle is to convert relatively inert carbonaceous dust from the ISM into the vital organic precursors to life such as amino acids and sugars intimately mixed with dust and ice in primitive planetesimals. Since the number of carbon atoms entering the Solar Nebula as dust exceeds the number of atoms entering the nebula as oxide grains, the formation of large quantities of complex organic molecules may represent the largest single chemical cycle in the nebula.
References:
Boynton W. V. 1985. In Protostars and Planets II. University of Arizona Press, pp. 772-787.
Bernatowicz T. J. and Cowisk R. 1997. In Astrophysical implications of the laboratory study of presolar materials. AIP Conference Proceedings, 402:451-474.
Ciesla F. L. and Sandford S. 2012 Science 336:452-454.
Clayton R. N. 2002 Nature 415: 860–861.
Clayton, R. Grossman, L. and Mayeda, T. 1973 Science 182:485.
Hill H. G. M. and Nuth, J. A. 2003 Astrobiology 3:291–304.
Jones A. P. and Nuth J. A. III 2011 Astronomy and Astrophysics 530:A44.
Lyons J. R. and Young E. D. 2005 Nature 435: 317–320.
McKeegan, K. D., Kallio, A. P. A., Heber, V. S., Jarzebinski, G., Mao, P. H., Coath, C. D. Kunihiro,
T., Weins, R. C., Nordholt, J. E., Moses, R. W., Reisenfeld, D. B., Jurewicz, A. J.G. and Burnett, D. S. 2011 Science: 332, 1528-1532.
Nittler L. R. et al. 1997. Astrophysical Journal 483:475-495.
Nuth J. A. et al. 2008. Astrophysical Journal Letters 673:L225–L228.
Nuth J. A. et al. 2000. Nature 406:275–276.
Yurimoto, H. and Kuramoto, K., 2004 Science 305, 1763 – 1766.
Publications
-
Johnson, N. M., Elsila, J. E., Kopstein, M., & Nuth, J. A. (2012). Carbon isotopic fractionation in Fischer-Tropsch-type reactions and relevance to meteorite organics. Meteoritics & Planetary Science, 47(6), 1029–1034. doi:10.1111/j.1945-5100.2012.01370.x
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Natasha Johnson
Co-Investigator
Joseph Nuth
Co-Investigator
-
RELATED OBJECTIVES:
Objective 1.1
Formation and evolution of habitable planets.