2010 Annual Science Report
 University of Wisconsin
Reporting | SEP 2009 – AUG 2010
University of Wisconsin
Reporting | SEP 2009 – AUG 2010
Project 2C: Computational Studies of Calcium Mineralization (Calcium Phosphate Nucleation and Dolomite Formation) as Model Systems for Biomineral Signatures on Earth and Other Solid Planetary Bodies
Project Summary
The unique morphologies of biominerals produced by organisms, when complemented by other chemical and isotopic signals, may serve as a potential biosignature for life on Mars and other solid planets. The mechanisms by which biology promotes organic-mediated biomineralization must be understood in order to distinguish them from look-alike minerals formed by the interaction of non-biological organic molecules or by inorganic physical-chemical processes. We have used the Molecular Dynamics and Bioinformatics computational chemistry approaches to determine the potential role of soluble proteins, commonly found in the organic part of biominerals, in controlling nucleation of the earliest solid Ca-PO4 inorganic precursor in hydroxyapatite (Ca5(PO4)3OH) biomineralization, and whether and how the conformation of a peptide (alpha-helix versus random coil) influences the nucleation pathway of hydroxyapatite compared to the inorganic system. We have found that the random coil conformation of the peptide promotes formation of an amorphous Ca-PO4 cluster, which ultimately transforms into crystalline hydroxyapatite. Identification of bioorganic molecule-promoted pathway of rapid amorphous solid formation, rather than direct mineral crystal templation, could help determine potential biomineral biosignatures on Mars and other solid worlds. Our project addresses NASA Astrobiology Institute’s (NAI) Roadmap goals of recognizing and preserving biosignatures and NASA’s Strategic Goal of advancing scientific knowledge of the origin and evolution of the Earth’s biosphere and the potential for life elsewhere.
Project Progress
The earliest hard exoskeleton-forming fossils are from the Vendian period (~ 650 – 543 Mya), also known as the Ediacaran period, in the latest Proterozoic Era, and just predating the Cambrian Explosion which is itself marked by a rapid increase in the occurrence of preserved mineralized fossils in the rock record. The earliest mineralized exoskeletons from the Vendian show a preference for calcium phosphate (apatite) mineralization but the dominant biomineral shifts to calcium carbonate in the Cambrain Explosion (Lowenstam and Weiner, 1989). Interestingly, the inarticulate brachiopods, including Lingula, are among the earliest to appear in the Cambrian record and are the only brachiopds to show calcium phosphate (francollite, which is fluorinated hydroxyapatite) mineralization, whereas all other and later brachiopods show calcium carbonate mineralization. Thus, studying the mechanisms of calcium phosphate mineralization may help understand the earliest evolution of exoskeletons and the preservation potential of fossils in the rock record. Vertebrates are characterized by internal skeletons made up of bone, which is a composite material of organic matrix and the calcium phosphate mineral, hydroxyapatite (Ca5(PO4)3OH, or dahlite whichis carbonated hydroxyapatite). Bone is well-studied, so we chose to use it as a model system for understanding the mechanisms of organic-mediated Ca-PO4 mineralization.
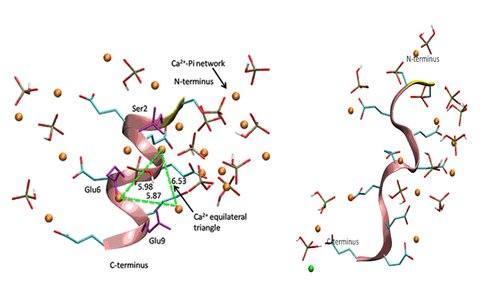
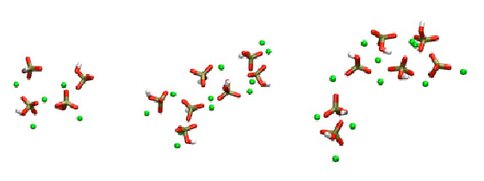
Bone sialoproteisn (BSP) is a highly phosphorylated, acidic, noncollagenous protein in bone matrix. Although BSP has been proposed to be a nucleator of hydroxyapatite (Ca5(PO4)3OH), the major mineral component of bone, no detailed mechanism for the nucleation process has been elucidated at the atomic level to date. In the present work, using a peptide model, we apply molecular dynamics (MD) simulations to study the conformational effect of a proposed nucleating motif of BSP (a phosphorylated, acidic, 10 amino-acid residue sequence) on controlling the distributions of Ca2+ and inorganic phosphate (Pi) ions in solution, and specifically, we explore whether a nucleating template for orientated hydroxyapatite could be formed in different peptide conformations. Both the α-helical conformation and the random coil structure have been studied, and inorganic solutions without the peptide are simulated as reference. Ca2+ distributions around the peptide surface and interactions between Ca2+ and Pi in the presence of the peptide are examined in detail. From the MD simulations, in some cases for the α-helical conformation, we observe that a Ca2+ equilateral triangle forms around the surface of peptide which matches the distribution of Ca2+ ions on the (001) face of the hydroxyapatite crystal (Figure 1 a). However, the template structure does not form consistently for various different starting ion distributions around the α-helix peptide, and in some simulations, even where the template does form, it is unstable over longer simulation periods greater than 5 ns up to 35 ns. The random coil conformation of the peptide helps to stabilize an interconnected Ca-PO4 network around the peptide which remains interconnected for the duration of the simulations (up to 35 ns) (Figure 2b), and this network more stable that the Ca-PO4 clusters formed in the inorganic, reference solution which tend to form and fall apart (Figure 2). Therefore, independent of conformations, the BSP nucleating motif is more likely to help nucleate an amorphous calcium phosphate cluster, which ultimately converts to crystalline hydroxyapatite.
These results are consistent with recent experimental evidence for amorphous Ca-PO4 precursor, which recrystallizes to francollite in Lingula anatina shell, and for amorphous CaCO3 precursors to calcite and aragonite biominerals. The consistency between our Ca-PO4 and previous Ca-CO3 studies indicates that universal principles underly biomineralization processes and the understanding gained from our computational study of peptide-promoted calcium phosphate nucleation can be extended to CaCO3 and other biominerals. The apparent switch in major biomineral from Ca-PO4 phases to Ca-CO3 phases at the end of the Vendian period may be related to the fact that the phosphate phases are more insoluble than the carbonates, so possibly, their formation may require a lower degree of regulation by organisms which were less evolved in the Vendian compared to the more evolved organisms of the Cambrian (Lowenstam and Weiner, 1989).
Future work will focus on the potential role of inorganic and organic ligands in promoting the formation of mixed cation carbonate such as dolomite ((Ca, Mg) CO3), and (Ca, Mg, Fe)CO3, which are known to form on Earth surface or near-surface conditions primarily through bacterial mediation.
Figure 1. Distribution of Ca2+ and HPO42- ions around the peptide in alpha-helix (a), and random coil (b) conformations, obtained by MD simulations using CHARMM force-field and 35 ns simulation time. In the alpha-helix structure, three Ca2+ ions associated with phosphoserine (Ser2) and glutamate (Glu6, Glu9) side-chains form a triangle of side length ~ 6 Å, which matches Ca2+ ion positions on the (001) face of HAP, which could act as a template to nucleate HAP (a). However, the triangle formed by the alpha-helix peptide is not stable and falls apart over a long, 35 ns simulation. The Ca2+ and HPO42- ions form a network around the peptide in random coil conformation (b), which is stable over the 35 ns simulation and is also more stable than Ca-PO4 networks formed in the inorganic, reference solution. Thus, the random coil structure of the peptide may promote nucleation of an amorphous, metastable Ca-PO4 precursor that converts eventually into crystalline HAP (Yang et al., 2010).
Figure 2. Examples of Ca2+ and HPO4 2- ion clusters in the inorganic, reference solution. These clusters are unstable, forming and breaking up several times over the course of a 35ns simulation (Yang et al., 2010).
Publications
-
Yang, Y., Cui, Q., & Sahai, N. (2010). How Does Bone Sialoprotein Promote the Nucleation of Hydroxyapatite? A Molecular Dynamics Study Using Model Peptides of Different Conformations. Langmuir, 26(12), 9848–9859. doi:10.1021/la100192z
- Yang Y., S.N. & Romanek, C. (In Preparation). Insight on dolomite formation – A computational study of Mg2+ dehydration in aqueous solution with presence of hydrogen sulfide. Geochim. Cosmochim. Acta.
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Yang Yang
Postdoc
-
RELATED OBJECTIVES:
Objective 7.1
Biosignatures to be sought in Solar System materials