2010 Annual Science Report
 Massachusetts Institute of Technology
Reporting | SEP 2009 – AUG 2010
Massachusetts Institute of Technology
Reporting | SEP 2009 – AUG 2010
Environmental Oxygen and the Rise (And Fall) of Metazoans
Project Summary
The viability of complex animals is intimately linked to the availability of molecular oxygen. This project looks at the environmental signatures of oxygen production and availability on a range of timescales and the connection between the stages of Precambrian oxygenation and biological complexity. Increasingly, it is becoming clear that the major mass extinction events of the Phanerozoic are strongly correlated to episodes of deoxygenation.
Project Progress
Environmental oxygen and the rise (and fall) of metazoans
The Bosak Laboratory at MIT investigated how microbial processes involving oxygenic photosynthesis leave morphological and isotopic imprints in sediments and discovered a formerly unrecognized fossil record of protistan life in carbonate strata that were deposited between the two global glaciations during the Neoproterozoic. In collaboration with Daniel Rothman, we show that the cm-scale spacing in modern conical stromatolites and some of the oldest stromatolites on Earth is a direct consequence of the photosynthetic activity of stromatolite-forming microbes, thus providing the first quantitative relationship between a microbial metabolism and preservable geometries of stromatolites (Petroff et al., 2010). Our laboratory experiments and field observations also demonstrate that vertical tubular microbialites can form when photosynthetic microbes colonize, move upward and grow outward on gas bubbles (Bosak et al., 2010a). Our experiments also question whether the conical shape of modern and ancient small stromatolites is directly related to microbial phototaxis and growth toward light (Liang et al., 2010). This work was presented in a lecture in the Science for the Public Series televised by WGBH/PBS. Our study of roll-up structures formed in carbonate strata immediately after the first Neoproterozoic global glaciation (Snowball Earth event) relate the previously enigmatic deformation of microbially-bound layers to the expulsion of fluids associated with the rapid precipitation of minerals (Pruss et al., 2010). These Neoproterozoic strata also contain abundant fossils of protists which survived the first Snowball Earth event (Bosak et al., 2010b). We are currently investigating this record in greater detail. In collaboration with Shuhei Ono (MIT), we show that the preference of a sulfate reducing microbe for light sulfur isotopes depends on its organic food source and how slowly this microbe grows. This finding (Sim et al., 2010) suggests that the highest sulfur isotope effects in sediments of all ages may be due to the presence of very slow-growing and starving microbes.
The Pearson group has been investigating the response of the marine nitrogen cycle to episodes of ocean anoxia in the Phanerozoic (specifically the Cretaceous), with the objective of focusing eventually on the Proterozoic. The nitrogen isotopic fractionation between biomass and chlorophyll was determined for a suite of prokaryotic and eukaryotic photosynthetic species (Higgins et al., GCA, 2010). Investigating a Cretaceous Ocean Anoxic Event (OAE 2) using these principles showed that eukaryotic phytoplankton remained an important component of the surface water primary producer community throughout the event (Higgins et al., submitted, 2010). The results suggest that extreme negative excursions in nitrogen isotopic values of biomass are an indicator of deep ocean redox state – specifically, the balance between reduced (ammonium) and oxidized (nitrite, nitrate) N species. This suggests that nitrogen in ancient sediments can reveal the state of the global redox balance and nitrogen cycle.
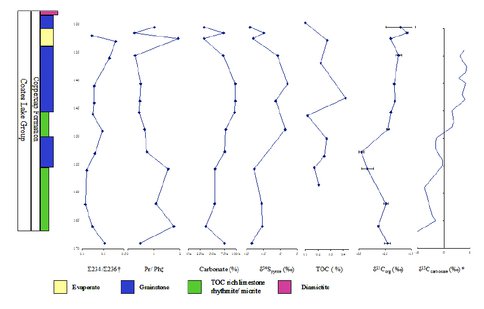
Katherine Thomas, a masters student working between the Ono, Bosak and Summons labs has conducted a biogeochemical study of the pre-Cryogenian Coppercap Formation. Sulfur and carbon stable isotope ratios and organic biomarker abundance were performed on drill core samples from the Coppercap Formation of the Coates Lake Group in the Windermere Supergroup of the MacKenzie Mountains to reconstruct an environmental condition proceeding the first Neoproterozoic Snowball Earth event. Based on aryl isoprenoids, n-propyl cholestane, isopropyl cholestane, total organic carbon, carbonate mineral analysis, δ34S from pyrite, δ13Corganic and δ13Ccarb values, environmental conditions of the Coppercap Formation were reconstructed. The presence of n-propyl cholestane throughout the Coppercap Formation indicate that marine pelagophytes were present throughout the section correlated with the theory of a lithospheric stretching event leading to the creation intracratonic oceans in the region. The Coppercap Formation was deposited under a largely euxinic environment evidenced by the presence of purple and green sulfur bacteria (aryl isoprenoids) and relative enrichment of δ34S values. Higher δ34S are correlated to reducing oceanic conditions and increased carbonate deposition (Figure 1). The presence of green sulfur bacteria indicates hydrogen sulfide rich waters, photic zone euxinia and the possibility of a persistent microbial mat throughout the deposition of the Coppercap Formation. Hydrogen sulfide rich waters in the deposition of the Coppercap Formation follow the theory of hydrogen sulfide rich waters persisted until later into the Cryogenian Period. Variations in the δ13Corg and the δ13Ccarb are correlated with a period of slightly oxic conditions indicating the presence of increased microbial activity present during relatively more oxic depositional conditions. Purple and green sulfur bacterial mats in the Coppercap Formation demonstrate the possibility for globally extensive euxinic conditions preceding the Cryogenian Period. Further studies should investigate the relationship between biogeochemical cycling and the lipid biological material of samples of comparable pre-Cryogenian rock formations to determine the geographical extent of euxinia in the Neoroterozoic.
Rothman’s group is studying ways in which the Earth’s carbon cycle would be perturbed at transitions from low oxygen to high oxygen environments. The late-Proterozoic rise of metazoans is likely one such transition. Another is the late Permian, during which a growing body of evidence, much of which is due to the Summons group-indicates that the oceans were anoxic. Earth’s greatest biotic crisis-the end-Permian extinction-may be related to the anoxic state. Recent work of the Rothman group suggests that the end-Permian extinction was accompanied by a major perturbation of the carbon cycle that was initially excited at least about 100 Kyr before the peak extinction activity. (The time scale arises from recent work in the Bowring group.) This new analysis, based on carbon-isotopic data in widely separated locations, suggests that the changes took the form of a dynamical instability with characteristics similar to a singular blow-up. These inferred dynamics suggests that an initially small biogeochemical perturbation, occurring at least about 100 Kyr before the extinction, led inexorably and predictably to the biosphere’s demise. The instability may have been triggered by an environmental perturbation due, e.g., to Siberian volcanism.
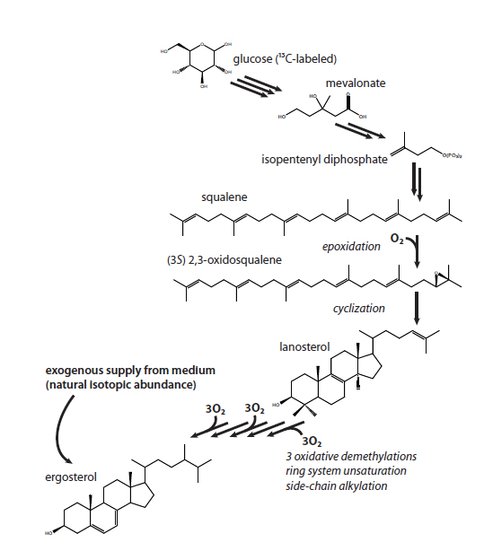
Steroidal hydrocarbons are proposed to be robust proxies for the availability of free oxygen in the environment (Sessions et al., 2009). This is because the biosynthesis of sterols is a highly oxygen-dependent process and earlier research based on enzyme assays (Jahnke and Klein, 1983) showing that the first oxygen-dependent step in the yeast pathway to ergosterol could be detected at oxygen levels as low as 0.03% (vol/vol) or the equivalent of micromolar concentrations of oxygen in water. Jacob Waldbauer (Waldbauer et a., 2010) has now shown that biosynthesis of 13C-labelled ergosterol can, in fact, be detected in yeast supplied with 13C-labeled glucose and as little as 7nM O2 using the approach depicted in Figure 1.
Modern conical photosynthetic biofilms made by oxygen-generating bacteria. The spacing between the individual vertical structures depends on the microbial activity during the day. Similar spacing can be recognized in conical structures as old as ~ 3 billion years. The field of view is ~ 5 cm across.
Small conical stromatolites from Nzuzi, South Africa (3.0-2.9 billion years old). The cm-scale of these structures implies that they formed in the presence of photosynthetic biofilms. The investigation of internal textures of these stromatolites will test whether some of these biofilms could have produced oxygen.
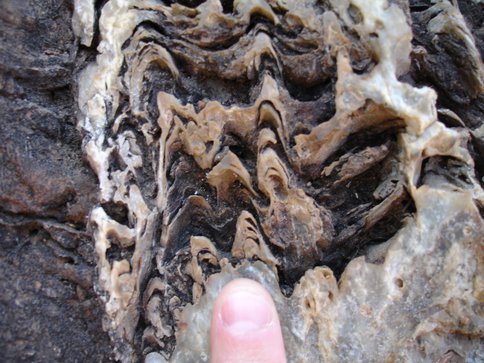
Microfossils in the laminae of Neoproterozoic cap carbonates from the Rasthof Formation, Namibia. The dark-walled microfossils are abundant within the laminae of fossilized microbial mats.
Vertical biofilm that grows around an oxygen-rich bubble. The more than 1 cm-tall tower grew within five hours after the onset of photosynthetic activity.
Figure 1. Biogeochemistry of the Cryogenian Coppercap Formation. Coppercap chemostratigraphy showing results for carbonate (), total organic carbon (), δ13C organic (‰), δ34Spyrite (‰) and biomarker analysis for pristane/ phytane (pr/ph), Σ2,3,4/ Σ2,36, of aryl isoprenoids and δ13C carbonate (‰). † Σ2,3,4/ Σ2,3,6 values are the sum of the 2,3,4 and 2,3,6 peaks from chromatography analysis of biolipids. Peaks represent the presence of green sulfur bacteria and purple sulfur bacteria, respectively. ‡ pr/ph value are the ratio of pristane to phytane; pr/ph >1 suggest more oxic conditions, pr/ph <1 represents anoxic conditions *δ13Ccarbonate values for this section are credited to F. MacDonald (unpublished).
Figure 2 Sterol biosynthesis in yeast. An abbreviated summary of the biosynthesis of sterols in yeast, showing the flow of carbon from glucose to ergosterol. The first oxygen-requiring step is the epoxidation of squalene; subsequently, O2 is also required for the removal of three methyl groups from the sterol ring system to produce the functional membrane lipid. In these experiments, cells can either synthesize sterols de novo (and hence with a 13C label) or take up unlabeled ergosterol from the medium.
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
David Johnston
Co-Investigator
Andrew Knoll
Co-Investigator
Shuhei Ono
Co-Investigator
Meytal Higgins
Doctoral Student
Alexander Petroff
Doctoral Student
Min-Sub Sim
Doctoral Student
Katherine Thomas
Graduate Student
-
RELATED OBJECTIVES:
Objective 4.1
Earth's early biosphere.
Objective 4.2
Production of complex life.