2013 Annual Science Report
 University of Wisconsin
Reporting | SEP 2012 – AUG 2013
University of Wisconsin
Reporting | SEP 2012 – AUG 2013
Project 1F: Organics Exposure in Orbit (OREOcube): A Next-Generation Space Exposure Platform
Project Summary
The OREOcube (ORganics Exposure in Orbit cube satellite) experiment on the International Space Station (ISS) investigates the effects of solar and cosmic radiation on organic thin films. By depositing organic samples onto inorganic substrates, structural changes and photo-modulated organic-inorganic interactions are examined to study the role that solid mineral surfaces play in the photo-chemical evolution, transport, and distribution of organics. The results of these experiments in low Earth orbit (LEO) allow extrapolation to different solar system and interstellar/interplanetary environments. Organic molecules appropriate for study in thin-film form include biomarkers such as amino acids and nucleobases, as well as polyaromatic hydrocarbons (PAHs), redox molecules, and organosulfur compounds. Inorganic substrates include silicates, metal oxides, iron sulfides, nano-phase iron, and iron-nickel alloys. By measuring changes in the UV-vis-NIR spectra of samples as a function of time in situ on the ISS, OREOcube will provide data sets that capture critical kinetic and mechanistic details of sample reactions that cannot be obtained with current exposure facilities in LEO. Combining in situ, real-time kinetic measurements with post-flight sample analysis will provide time-course studies, as well as in-depth chemical analysis, enabling us to characterize and model the chemistry of organic species associated with mineral surfaces in the astrobiological context.
Project Progress
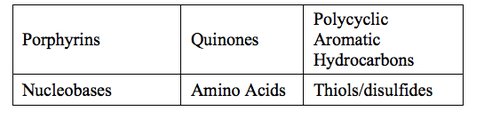
In preparation for the OREOcube experiment, we have performed laboratory characterizations of five classes of candidate organic compounds (see Table 2) in combination with inorganic catalysts. Amino acids, the building blocks of proteins, are generally the most abundant single class of molecules in bacterial cells followed by nucleobases (associated with DNA and RNA). Both represent important target molecules in the search for life. PAHs are abundant and ubiquitous in galactic and extragalactic regions, protoplanetary disks, and solar system objects. Their spectral measurements indicate the presence of a large variety of individual compounds, clusters, and PAH-related molecules. A class of redox-active aromatic molecules that play roles in biological electron transfer processes, the quinones, has been formed in numerous laboratory simulations of UV-photolyzed interstellar and planetary ices containing PAHs.
Representative compounds from four of the organic sample classes (porphyrins, quinones, PAHs, amino acids) listed in Table 1 have been investigated during the Organism/Organic Exposure to Orbital Stresses (O/OREOS) Mission. Three of these compounds isoviolanthrene (IVA; PAH class), iron-5,10,15,20-tetraphenyl-21H,23H-porphine iron(III) chloride (FeTPP; porphyrin class), and 1,5-dihydroxyanthraquinone (anthrarufin; quinone class) will be used as OREOcube samples. Anthrarufin is a partially oxidized form of 9,10-anthraquinone, which is present in plants, fungi, lichens, and insects. FeTPP is representative of a large class of redox-active organometallic compounds, typified by porphyrins and phthalocyanines, that enable biological processes from photosynthesis to respiration. By using these samples in OREOcube inorganic catalysis experiments, O/OREOS science will be expanded. Additionally, OREOcube will provide data sets which will be used to examine the dependence of reaction rates and products on parameters related to ISS and O/OREOS satellite orbit, primarily the time-varying pattern of light/dark exposures and temperatures.
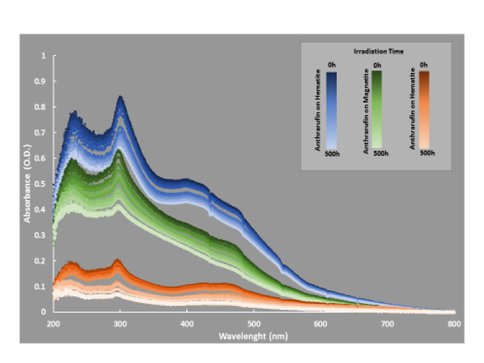
We present here results from experiments conducted with iron oxide thin films in combination with a thin film of the quinone anthrarufin. Iron oxide films were prepared for laboratory studies by physical vapor deposition of reduced iron in a high-vacuum deposition chamber (~ 1 × 10-4 Pa). After deposition, the films were converted to magnetite or hematite by heating for 4 h, in the presence of oxygen atmosphere, at 175 °C or 575 °C, respectively. The thickness of the iron films prior to oxidation was 2 nm. Anthrarufin (51 nm) thin films were deposited as described above either on uncoated fused silica (SiO2) or on top of hematite or magnetite deposited on SiO2. Anthrarufin (Sigma-Aldrich, St. Louis, MO, USA) was pre-purified by heating under vacuum prior to film deposition. UV exposures were performed using a Hanovia 1000 W Xe arc lamp with an IR water filter to prevent sample heating, providing an integrated (200 – 400 nm) UV flux of 62 ± 0.5 W/m2.
The sample-coated windows were not sealed into sample cells, but instead the inorganic/organic films were exposed, as a single lot, in sample chamber with a Suprasil fused silica optical port. A 800 Pa CO2 (99.99%) atmosphere, with a mass flow controlled 0.15 sccm flow rate, was maintained using an oil-free diaphragm pump. The dewpoint was held between -50° and -60° C. The overall objective of these experiments was to examine UV photolysis rates under Mars-relevant conditions (i.e., Mars pCO2 and pH2O in combination with iron oxide catalysis) as a selection criterium for samples for the OREOcube ISS experiment (see Figure 1).
The results of Figure 1 highlight the power of time-resolved spectral measurements that record not only starting and ending points, but also dynamic changes and alteration rates. For OREOcube on the ISS, the same sort of kinetic analysis will be possible, but the time between measurements will be more regular and there will be no intervening air exposures; the experiment will be fully autonomous and remotely controlled. Our pre-flight simulation experiment with magnetite and hematite thin films demonstrates that kinetic differences in the photolyzed samples are measurable. Hematite with its known semiconducting photo-catalytic properties showed the most rapid decrease in integrated absorbance due to anthrarufin degradation while anthrarufin alone showed the slowest photodegradation.
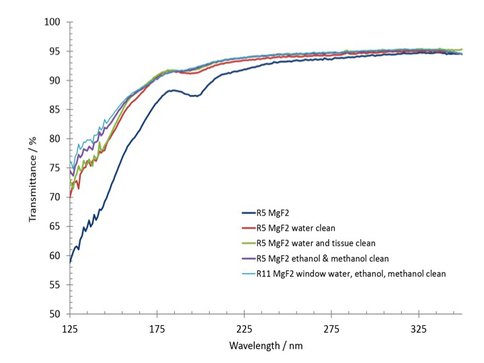
In addition to pre-flight experiments, baseline flight and ground control samples were prepared with organic thin films of isoviolanthrene, iron tetraphenylporphyrin and anthrarufin on various catalytic substrates including Cr2O3, Fe-Ni, TiO2 and Fe3O4 and Fe2O3. All films were deposited onto magnesium fluoride (MgF2) windows, which were characterized prior to film deposition by UV-vis spectroscopy. Window cleaning procedures were tested on a subset of MgF2 windows and measured in the far-UV region at the ASTRID synchrotron facility in Aarhus, Denmark (see Figure 2).
All flight samples have been sealed within flight sample cells containing argon. Baseline UV-Vis spectra have been recorded while Argon leak rate testing of all flight cells is still ongoing. Flight samples will be launched (anticipated in 2015) to and installed on the outside of the International Space Station (ISS).
Publications
-
Ehrenfreund, P., Ricco, A. J., Squires, D., Kitts, C., Agasid, E., Bramall, N., … Young, A. (2014). The O/OREOS mission—Astrobiology in low Earth orbit. Acta Astronautica, 93, 501–508. doi:10.1016/j.actaastro.2012.09.009
-
Elsaesser, A., Quinn, R. C., Ehrenfreund, P., Mattioda, A. L., Ricco, A. J., Alonzo, J., … Santos, O. (2014). Organics Exposure in Orbit (OREOcube): A Next-Generation Space Exposure Platform. Langmuir, 30(44), 13217–13227. doi:10.1021/la501203g
-
PROJECT INVESTIGATORS:
-
PROJECT MEMBERS:
Pascale Ehrenfreund
Project Investigator
Andreas Elsaesser
Collaborator
Richard Quinn
Collaborator
Antonio J. Ricco
Collaborator
Farid Salama
Collaborator
-
RELATED OBJECTIVES:
Objective 3.1
Sources of prebiotic materials and catalysts
Objective 5.3
Biochemical adaptation to extreme environments
Objective 7.1
Biosignatures to be sought in Solar System materials