
The occurrence of surface water on present-day Mars is increasingly supported by data from orbiting spacecraft and landed rovers, as well as theoretical studies. Pure liquid water is not stable in the extremely cold and dry surface conditions on Mars; however, concentrated salt solutions can allow liquid water to form. Salt solutions can depress the freezing point of liquid water down to -75°C or lower due to supercooling, and hygroscopic crystalline salts can spontaneously absorb water from the atmosphere to form brine. In this talk, I will explore what evidence currently supports the formation of liquid water on Mars, what the salt composition of that water might be, and the potential for life in such brines.
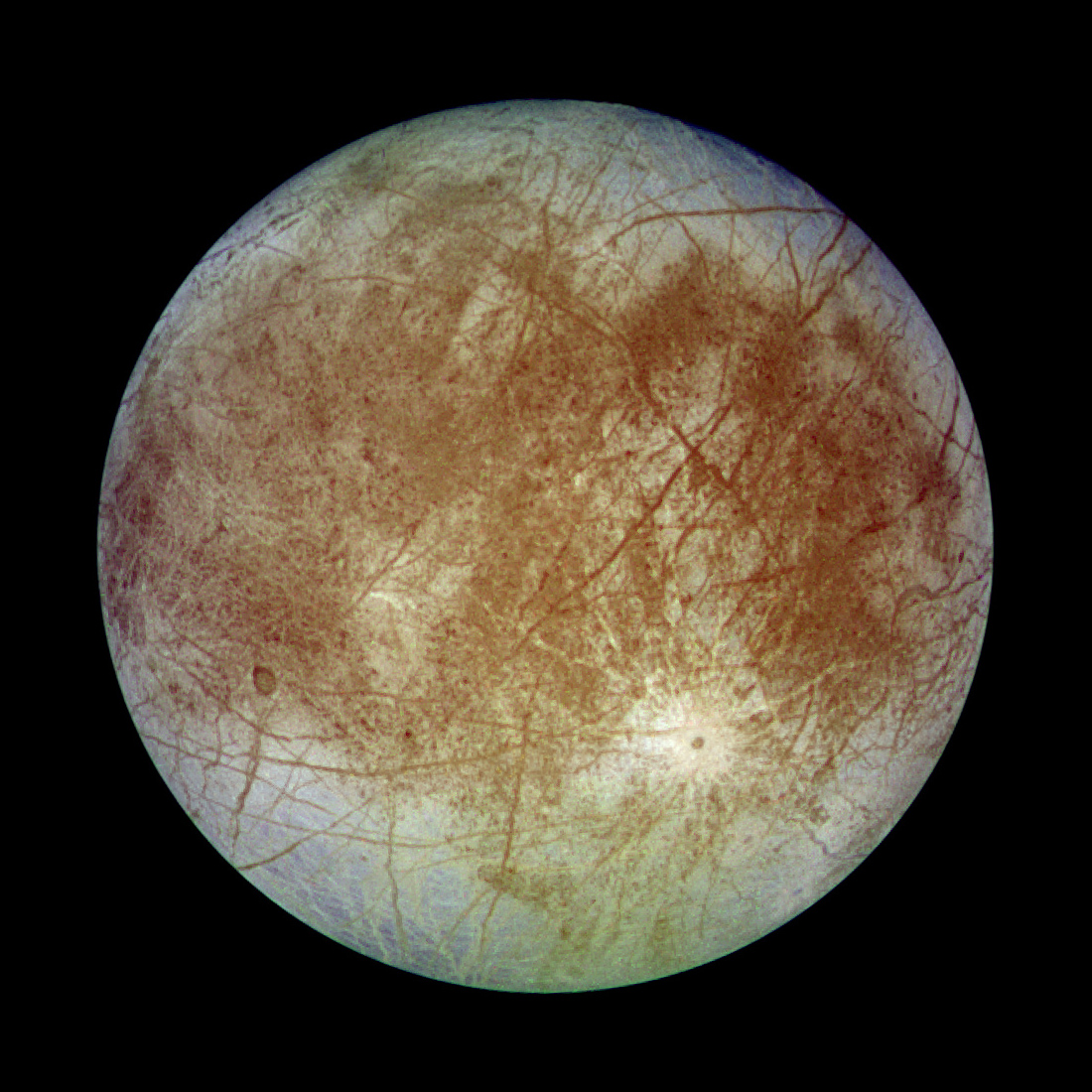 Getting Under Europa’s Skin
Getting Under Europa’s Skin Tracing Formation and Evolution of Outer Solar System Bodies Through Stable Isotopes and Noble Gas Abundances
Tracing Formation and Evolution of Outer Solar System Bodies Through Stable Isotopes and Noble Gas Abundances Photosynthesis, a Planetary Revolution
Photosynthesis, a Planetary Revolution Xenon: King of the Gases
Xenon: King of the Gases